Background: Metastatic breast cancer (MBC) patients have numerous options for treatment. However, it is essential to consider treatments with favorable toxicity profiles and convenient modes of administration. Eribulin has shown effectiveness in aggressive MBC, but there is a lack of sufficient real-world data specific to Indian patients.
Patients and methods: We conducted a retrospective audit of patients with MBC who received intravenous Eribulin between 2019 and 2023 at a dosage of 1.4 mg/m2 on days 1 and 8 every 3 weeks. The median Progression-free survival (PFS) and overall survival (OS) were estimated using the Kaplan-Meier method.
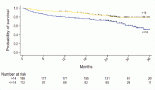
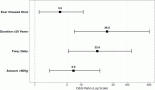
Results: During the specified time, 107 consecutive patients with MBC received Eribulin treatment. The median age was 52 years (range, 28–75 years) with 3 patients with male breast cancer. The median number of prior chemotherapy lines and involved sites were 3 (range, 2–5) and 3 (range, 1–6), respectively. Visceral involvement was present in 84 (78.5%) patients. A median of 3 cycles of Eribulin (range, 1–11) was administered. Eribulin resulted in partial responses in 49 (45.8%) patients, stable disease in 11 (10.3%) patients and progressive disease in 47 (43.9%) patients. The median PFS was 4.0 months (95% CI: 3.4–4.6), and the median OS was 10.0 months (95% CI: 8.3–11.7). For patients with triple-negative breast cancer (TNBC), the median OS was 8 months (95% CI: 5.6–10.4), whereas non-TNBC patients had a median OS of 11 months (95% CI: 9.1–12.8) (hazard ratio, 1.9, 95% CI: 1.2–3.1, p = 0.002). Eribulin was well-tolerated, with dose reduction was needed in 9 (8.4%) of the patients in the overall cohort.
Conclusion: Eribulin is a viable and safe option for treating heavily pre-treated MBC in real-world settings. The study found comparable efficacy in both TNBC and non-TNBC patients.