Prospective evaluation of locoregional control in oral cavity squamous cell carcinoma with lower or mid infratemporal fossa involvement treated with surgery and adjuvant concurrent chemoradiotherapy
Prasoon Mishra1, Rahat Hadi1, Ajeet Kumar Gandhi1, Madhup Rastogi1, Rohini Khurana1, Ashish Singhal2, Surendra Prasad Mishra1, Anoop Srivastava1, Avinav Bharati1, Ashish Chandra Agarwal3, Avinash Poojari1, Vachaspati Kumar Mishra1, Raunaq Puri1, Akanksha Manral1, Vikas Gupta1, Bhoopendra Pratap Vishwaranjan1 and Saumyta Mishra4
1Department of Radiation Oncology, Dr. Ram Manohar Lohia Institute of Medical Sciences, Lucknow 226010, India
2Department of Surgical Oncology, Dr. Ram Manohar Lohia Institute of Medical Sciences, Lucknow 226010, India
3Department of Otorhinolaryngology, Dr. Ram Manohar Lohia Institute of Medical Sciences, Lucknow 226010, India
4Department of Surgical Oncology, Mahavir Cancer Sansthan, Patna 801505, India
Abstract
Purpose: Oral cavity squamous cell carcinoma (OCSCC) is the most prevalent malignancy of the head and neck malignancy in India. In locally advanced OCSCC, a combination of definitive surgery followed by postoperative radiotherapy (RT), with or without concurrent chemotherapy, offers superior oncologic outcomes compared to definitive chemoradiotherapy alone. The prognosis of T4b OCSCC with infratemporal fossa (ITF) involvement largely depended on the degree of invasion. Tumours confined to the lower or mid ITF tend to have relatively better outcomes than those with extensive or high-level involvement. This study prospectively evaluates the treatment outcomes in patients with OCSCC involving the lower or mid ITF who underwent curative surgical resection followed by adjuvant concurrent chemoradiotherapy (CCRT).
Methods: This is a single-arm prospective interventional study between January 2021 and October 2022, which included 20 patients of clinically T4b OCSCC as per the American Joint Committee on Cancer (8th edition) with lower or mid ITF involvement. Patients with tumour invasion into the skull base, carotid artery, prevertebral fascia or pterygomaxillary fissure were excluded to maintain a uniform study cohort. All patients were treated with curative intent with surgery include ITF clearance followed by adjuvant CCRT. Primary endpoint was to evaluate loco-regional control (LRC) and secondary end point was to assess acute toxicities according to the Common Terminology Criteria for Adverse Events v5.0. Kaplan-Meier survival analysis was done for assessing disease free survival (DFS), overall survival (OS).
Results: Median follow-up was 11.5 months (4–21 months). The median age was 39 years (range 32–67) with 95% males. Buccal mucosa was most common site with 19 patients (95%). All patients were clinically cT4b (Stage IV) stage involving low/mid ITF involvement with low ITF were 15(75%) and mid ITF patients were 5(25%) patients. All patients underwent definitive ITF clearance and received adjuvant CCRT with weekly concurrent cisplatin 35–40 mg/m2 to a median number of 6 cycles (5–7 cycles). Median RT dose was 64 Gray (60–66). 08 (40%) patients had grade 3 oral mucositis and odynophagia each. Skin toxicity and oral pain was present in 6(30%) and 5(25%) patients, respectively. Grade 4 toxicities were not observed. LRC at 3 months was 73.68% with 4 local and 1 locoregional failures only. LRC at 1 year was 36.8% with 8 local, 2 local-distant, 1 loco-regional-distant and 1 distant failure only. Median DFS was 10.4 months and median OS was not reached; four patients died. LRC at 3 months was better for patients with low ITF disease as compared to mid ITF disease (85.7% versus 40% (p = 0.04) and at 1 year was 42.8% versus 40% (p = 0.50).
Conclusion: Locally advanced OCSCC with low ITF involvement may benefit from tri-modality therapy of surgery with ITF clearance followed by adjuvant CCRT. The acute toxicity observed in study was within acceptable limits.
Keywords: low and mid infratemporal fossa involvement, ITF clearance, concurrent chemoradiotherapy, loco-regional control, locally advanced OCSCC
Correspondence to: Prasoon Mishra
Email: mishraprasoon28@gmail.com
Published: 19/03/2026
Received: 26/06/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Background
In India, head and neck cancers represent major health issue, contribute nearly 30% of all new cancer case in which lip and oral cavity constitute approximately 140,700 new cases. Oral cavity carcinoma is the most common subtype among males, mainly linked to the widespread use of tobacco in multiple forms. Its incidence is approximately 10.3% and it is a 2nd most common leading cause of cancer deaths (8.7%) [1, 2]. Over 50% present with advanced-stage disease, with gingiva-buccal sulcus complex cancers being the most prevalent [2].
Surgery is the primary treatment for oral cavity squamous cell carcinoma (OCSCC), with adjuvant radiotherapy (RT) with or without chemotherapy recommended for high-risk features such as advanced stage, positive margins or perineural and lymphovascular invasion (LVI). The optimal RT dose, fractionation and concurrent chemotherapy remain complex and are addressed through risk stratification frameworks [3–5]. The EORTC 22931 and RTOG 9501 trials established the foundation for the risk-based selection of adjuvant RT and concurrent chemoradiotherapy (CCRT) in postoperative OCSCC [6, 7].
These studies identified an intermediate-risk group between low-risk patients, who may not need adjuvant therapy and high-risk patients requiring adjuvant CCRT. This group includes pT1-4 tumours with close margins (1–5 mm), perineural invasion (PNI), LVI, involved nodes or poor differentiation.
Head and neck surgeons faces challenges in locally advanced OCSCC especially when the disease involves the infratemporal fossa (ITF) and are associated with a median survival of approximately 6 to 9 months [2, 8]. Advanced oral cavity cancers with ITF involvement are classified as T4b disease and designated as ‘very advanced disease’ in American Joint Committee on Cancer (AJCC) 8th edition staging system [9].
The ITF is a structure situated deep to the mandibular ramus and below the zygomatic arch. It contains the pterygoid muscles, V3, chorda tympani, otic ganglion, maxillary artery and pterygoid venous plexus. The masseter muscle is typically excluded [10–13].
ITF involvement is described as category low, intermediate or high based on anatomical spread, from local muscle involvement to extension into the intracranial compartment. Low and intermediate are operable with good outcomes, while high ITF, has variable prognosis [14–16].
The anatomy of the ITF and masticator space (MS) complicates cancer evaluation. Radiological assessment is challenging, and trismus limits clinical examination [17–19]. Due to neurovascular structure in the MS complete resection of tumour is very difficult.
cT4b oral cavity cancers are traditionally inoperable and managed palliative with radiation and chemotherapy or both [20, 21]. Due to complex anatomy ITF tumours were rarely resected but recent studies show improved outcomes with compartment resection especially below the mandibular sigmoid notch [15, 22–26]. In few studies neoadjuvant chemotherapy (NACT) has been used to downstage tumours and improve resectability [27–29].
cT4b carcinoma of oral cavity has poor outcomes. Patient with minimal involvement of ITF (mid-lower 1/3) may have better outcome as compared to other cT4 OCSCC patient if treated aggressively with surgery and adjuvant CCRT [10].
We aim to evaluate patients of OCSCC with minimal ITF (mid-lower 1/3) involvement treated with surgery followed by adjuvant CCRT administered as per our institutional protocol in view of the aggressive nature of ITF involvement in terms of loco-regional control (LRC) and acute toxicity of treatment.
Methods
Study design
20 patients of cT4 OCSCC (as per AJCC 8th edition) with low or mid ITF involvement were included in this single arm prospective interventional study from January 2021 to October 2022. All patients underwent curative surgical resection with standardized ITF clearance followed by, adjuvant CCRT was administered to all patients. No patients in this cohort received NACT prior to surgery.
The study was approved by the institutional ethics committee (IEC No.166/20/RMLIMS/ 2020) and written informed consent was taken from all patients prior to the enrolment.
Eligible patients had OCSCCs (except lip), aged 18–80 years, of either sex with a Karnofsky performance score ≥70, ITF Involvement mid and lower defined on cross-sectional imaging (contrast-enhanced CT or MRI) as Low ITF involvement (infranotch) on imaging as tumour localized below the mandibular (sigmoid) notch, structures involving such as the medial pterygoid, masseter or retroantral fat and Mid ITF involvement (supranotch) was defined as tumour extending superiorly above the notch with involvement of lateral pterygoid, temporalis insertion or upper pterygoid plates, without involving skull base or intracranial extension [15, 16, 30],with pathologically complete and appropriate surgical resection of primary disease and nodes with 1 or more of the following risk factors for adjuvant treatment: pT3/pT4, ≥pN2a, close margin (1–5 mm), PNI, lymphovasular invasion and depth of invasion (> 4 mm for tongue and ≥7 mm for buccal mucosa) [4, 31], high grade tumour, adequate bone marrow reserve, non metastatic disease, as per institutional protocol, ITF involvement itself was considered a high-risk feature; therefore, all patients were planned for adjuvant CCRT irrespective of classical high-risk factors such as margin positivity or extra capsular extension as stated in landmark study (category 1 as per NCCN) [6, 7].
Excluded patients were non-squamous cell carcinoma histology, high ITF involvement extending to the temporal fossa or infiltration into the pterygomaxillary fissure, carotid artery encasement or prevertebral fascia. Cases of T4b disease except for isolated ITF involvement were also excluded. Additional exclusions included patients with synchronous or metachronous second malignancies, metastatic disease, pregnancy, lactation or active infections such as HIV, Hepatitis B or Hepatitis C. Patients indicated for chemo-radiation but deemed unfit due to co morbidities, poor general condition or unwillingness to undergo chemotherapy were also excluded from the study.
Clinical evaluation
All patients were staged in accordance with the criteria outlined in the 8th edition of the AJCC Cancer Staging Manual, published in 2017. A thorough medical history and physical examination were conducted, along with routine hematological, renal and hepatic function tests, post-operative histopathological reporting, necessary CECT/MRI of the face and neck, chest X-ray (P/A view), abdominal ultrasonography and glomerular filteration rate assessment when clinically indicated.
Surgery
All patients underwent a standardized surgical procedure for ITF compartmental clearance, involving bite composite resection with comprehensive neck dissection (modified neck dissection type II/III based on nodal burden). Complete clearance was achieved by removing all muscles, fibro-fatty tissue, neurovascular bundles, retroantral fat and pterygoid plates within the specified region, adhering to the compartment surgery concept of removing the whole anatomical subunit encompasses the disease. Removal of the buccal branches of the facial nerve is required in advance diseases, if feasible upper division of the facial nerve and the marginal branch has to be preserved. Segmental mandibulectomy is commonly performed due to bone involvement or significant paramandibular spread, with reconstruction typically done using a bipaddled pectoralis major myocutaneous flap. All patients received adjuvant treatment as per surgical histopathology report [24].
Patients received oral hygiene counseling, dental prophylaxis with fluoride application, instructions for using oral rinses (benzydamine, sodium bicarbonate, saline gargles), clotrimazole lozenges during RT and their weight was recorded prior to treatment, with written informed consent. Gap between surgery and start of RT was kept ≤6 weeks.
Radiotherapy
Simulation and planning
CT simulation with contrast enhancement was performed using a 16-slice CT simulator (Siemens Somatom Sensation) with patients in a supine position; neck extended and immobilized using a carbon fiber base plate and a thermoplastic U-type extended face mask. Contrast enhanced images were acquired with a 3 mm slice thickness.
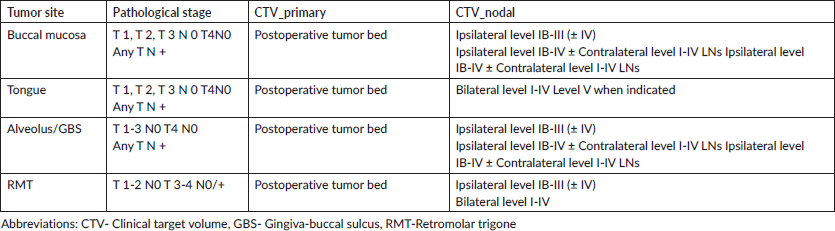
CT images were imported to the MONACO SIM (Version 5.11.03) contouring station via DICOM 3.0. Target volumes and organs at risk were contoured according to ICRU 50 & 62 guidelines. Clinical target volumes (CTVs) included the tumour bed and nodal regions, with a uniform 5 mm margin added to generate planning target volume, as per institutional protocol. The volumes taken have been presented in Table 1 [32, 33]. The data were then transferred to the XIO (Version 5.0) treatment planning system for further planning.
Dose prescription
RT was administered in two sequential phases, delivering a total dose of 60–66 Gray (Gy) in 30–33 fractions (2Gy per fraction), administered on weekdays (Monday through Friday) over a period of approximately 6 to 6.5 weeks. Patient was treated with three-dimensional conformal RTeither with parallel opposed anterior—posterior oblique field or bilateral parallel opposed lateral field matched with low anterior neck field (LAN) in 2 phases Figure 1 (1a–1d) via 6 MV photons along with concurrent chemotherapy with weekly Cisplatin 40 mg/m2. In the first phase, treatment was delivered using two parallel-opposed lateral photon fields matched with LAN field to encompass the bilateral face and neck including the lymphatic drainage areas to the dose of 44 Gy in 22 fractions. In the second phase, the cord is off by shifting the posterior border anteriorly and removing the LAN field followed by treating the target volume to the total dose of 60–66 Gy by bilateral parallel-opposed lateral fields. A representative dose colour wash distribution is illustrated in Figure 1. If clinically indicated and technically feasible, parallel opposed oblique fields were used to deliver dose up to 60–66 Gy to the Ipsilateral face and neck Figure 1 (2a–2d). Treatment plans were assessed using dose-volume histograms, planar and 3D isodose displays slice by slice and modified accordingly.
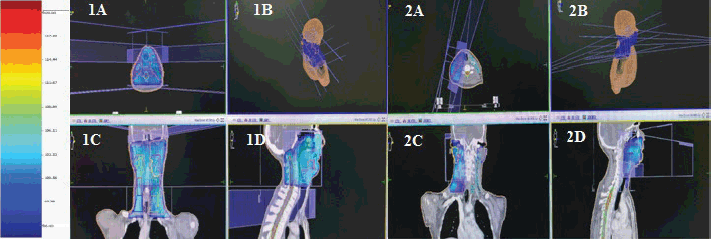
Figure 1. (1a–1d): Radiation treatment planning images of target volume delineation and dose distribution with parallel opposed lateral field matched with LAN. (2a–2d): Ipsilateral parallel oblique field.
Table 1. Description of CTVs taken for different sites of oral cavity.
Organ-at-risk dose constraints included a spinal cord Dmax ≤ 45 Gy and PRV Cord Dmax ≤ 50 Gy. Position verification was done by Electronic Portal Imaging Device twice weekly in the first week and then once weekly. Treatment was administered using a source-to-axis distance technique with 6 MV X-ray beams from a Linear Accelerator (ELEKTA Infinity/Synergy, Crawley, UK) with a 1 cm collimator leaf width at the Isocentre.
Patients were assessed weekly during RT with body weight recorded before and during treatment using a standardized weighing machine. Acute radiation toxicities including oral mucositis (OM), skin toxicity, odynophagia, dysphagia, oral pain and hematological toxicities were graded per Common Terminology Criteria for Adverse Events v5.0 [34] from the start of RT to 90 days post-treatment. Oral hygiene, fungal infections and nutritional status were also assessed weekly.
Post treatment follow up
Post-treatment completion, visits began 4 weeks after RT completion, followed by monthly visits for the first year and bimonthly for the second year. A minimum 6-month follow-up was conducted with documentation of toxicities. Locoregional status was evaluated at each visit of follow up with imaging (CECT/MRI/PET-CT) at 3 months after completion of RT to assess LRC. During subsequent follow-up, clinical examination was performed at every visit (monthly during the first year and bimonthly during the second year). Imaging was not performed at every visit but was repeated selectively at 3-month intervals in the first year when clinically indicated or earlier if new symptoms or suspicious findings were detected on clinical examination.
Statistical analysis
Statistical analysis was done with Statistical Package for Social Sciences (v25). Quantitative data were summarized with median, mean and standard deviation. LRC was defined as the absence of local and/or regional failure Locoregional failure defined as biopsy proven disease in postoperative bed or cervical nodes or both. LRC rates were calculated using crude proportions and comparisons between groups were descriptive without formal statistical testing. Disease free survival was measured from date of surgery till date of locoregional failure, distant metastasis or disease-related death, while overall survival (OS) was calculated from date of registration to the date of death from any cause. Survival rates were calculated using Kaplan–Meier method. The patients who were alive at the time of data cut off time or lost to follow-up were censored for survival analysis.
Results
Patient characteristics have been summarized in Table 2. Median follow-up was 11.5 months (4–21) months. All 20 Patients have completed the treatment. All patients underwent a standardized surgical procedure for ITF clearance with median number of lymph node dissected is 29 (16–61), only one patient with less than 18 nodes dissected. 17 patients had unilateral neck dissection (01 patients had supraomohyoid neck dissection, 19 had modified radical neck dissection and 1 had radical neck dissection) and 3 had bilateral neck dissection. 01 (05%) Patients had segmental mandibulectomy and 14 (70%) had hemi-mandibulectomy. 05 patients had upper alveolectomy. 12 out of 20 patients received unilateral RT and the remaining 08 received bilateral RT. All patients were pathologically pT4b (Stage IV) with ITF involvement. Median RT dose was 64 Gy (60–66 Gy). The median time interval between surgery and beginning of RT was 41 days (range: 30–49). The median overall treatment time defined as the interval from the date of surgery to the completion of postoperative RT was 107 days (75–123 days). The median duration of radiation therapy was 46 days (range: 35–52). The treatment compliance was good with all the patients receiving the full doses of RT as planned. All patients had infused concurrent chemotherapy with weekly cisplatin at a dose of 35–40 mg/m2, administered for a median of six cycles (range: 5–7). Out of 20 patients 9 patients (45%) received a reduced dose of 35 mg/m2 per week due to borderline renal function or prior grade 2 hematological toxicities. Dose reductions were made as per institutional safety protocols to maintain tolerability.
Table 2. Patient and tumour characteristics.
Toxicity analysis
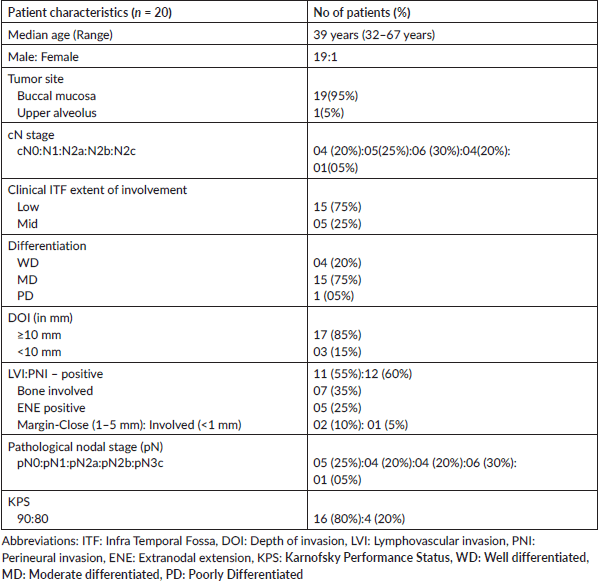
Figure 3 demonstrates the weekly progression of acute toxicities during the treatment. On weekly analysis, Grade 2 OM was observed in 12 patients (60%) by the 6th week, with Grade 3 confluent OM appearing in the third week. Number of patients with Grade 3 or higher toxicities increased as treatment progressed, reaching 8 patients (40%) by the end of the 6th week, with no Grade 4 toxicity.
Grade 2 skin toxicities were observed in 8 patients (40%) and Grade 3 in 6 patients (30%) by the end of 6th week. Grade 2 and 3 dysphagia started appearing during the 2nd and 3rd weeks of treatment, with Grade 3 seen in 8 patients (40%) by the end of 6th week and 11 patients (55%) had clinically Grade 2 toxicities with altered diet.
Grade 2 oral pains started appearing during the 2nd and 3rd weeks of treatment, while Grade 3 oral pain began in the 3rd and 4th weeks. By the end of treatment, Grade 3 toxicity was observed in 5 patients (25%) at the 6th week and Grade 2 toxicities were present in 10 patients (50%) at the 6th week.
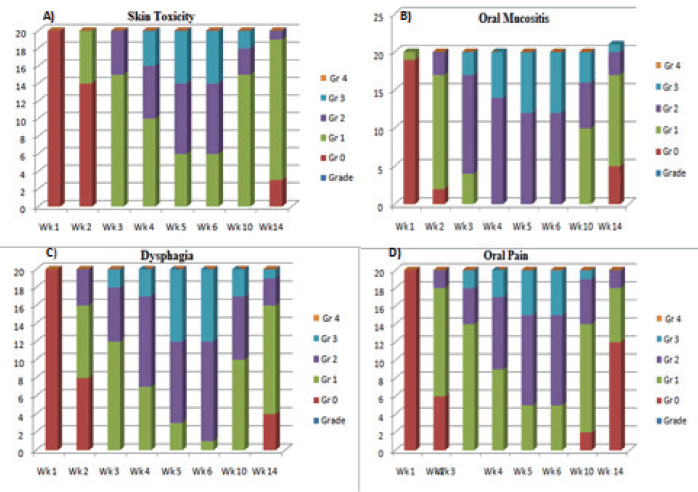
Figure 3. Stacked bars illustrating the temporal progression of acute toxicities for (a): Skin toxicity, (b): OM, (c): Pharynx/esophageal toxicity and (d): Oral pain.
Overall 8 patients (40%) had Grade 3 OM and odynophagia each, while 6 patients (30%) had Grade 3 skin toxicity and 5 patients (25%) had oral pain, respectively, with no Grade 4 toxicities were observed.
Two patients (10%) developed chemo-RT-induced Grade 2 anemia toxicities and 2 patients (10%) had Grade 2 neutropenia, with no patients experiencing thrombocytopenia. Overall toxicity evaluation 90 days after completion of RT showed OM and odynophagia in 1 patient (5%), with no skin toxicity or oral pain.
The median weight loss was 3 kg (range 1–5 kg), with 9 patients (45%) receiving IV fluids and 10 patients (50%) requiring tube feeding during RT. The median duration of enteral feeding was 20 days (range 12–40 days), with the median initiation time being 20 days (range 16–28 days) from the start of RT. Most patients were able to restart oral feeds within 5 weeks of completing RT.
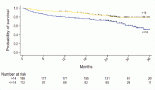
On clinical assessment at 1st month follow up 19 patients were without recurrence and one patient died due to covid pneumonitis. LRC at three month was 73.68% (14/19) with 4 local and 1 locoregional failures only. Five were given Palliative Chemotherapy. LRC at 1 year was 36.8% (7/19) with 8 local recurrences alone and 4 patients with distant metastasis were observed, 3 distant metastasis patients with loco and/or regional relapse and 1 patient had only distant metastasis. Figure 4 depicts Kaplan-Meier curve with Median disease free survival (DFS) of 10.4 month, however Median OS was not reached; four patients died: 2 due to primary disease, 1 due to distant metastasis in lung with loco-regional disease and 1 death related to other cause (Covid pneumonitis). Figure 5 depicts Kaplan-Meier curve of OS. The 1-year DFS rate and OS rate was 53% (95% CI: 34%–83%) and 74% (95% CI: 56%–96%). At 3 months, LRC was better in patients with low ITF disease compared to those with mid ITF involvement (85.7% versus 40%, p = 0.04); at one year, the difference narrowed (42.8% versus 40%, p = 0.50).
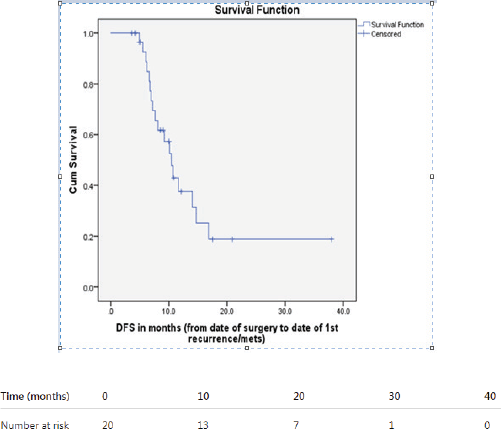
Figure 4. DFS curve.
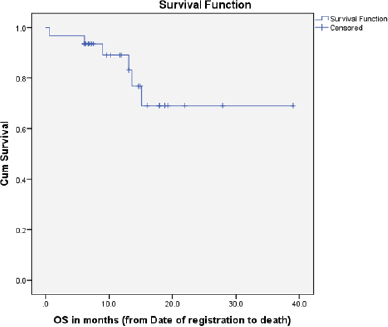
Figure 5. OS curve.
Discussion
T4b OCSCC with ITF involvement treatment remains complex due to challenging anatomy and limited resectability. Recent evidence supports curative surgery with ITF clearance followed by adjuvant chemoradiotherapy. Our prospective study focused on such patients with low and mid ITF involvement and demonstrated that this multimodality approach is feasible, tolerable and may offer improved short-term control.
In our study, adjuvant CCRT was administered to all patients irrespective of margin or extranodal extension (ENE) status, as ITF involvement itself was considered a high-risk feature in line with our institutional practice. Few studies historically showed poor outcomes of T4b OCSCC with MS or ITF spread, where surgery alone is not sufficient to achieve good outcome. Prior reports, including those by Trivedi et al [24] and Katna et al [30], have similarly depicted the aggressive biology of ITF-involved tumours and warrant for intensified adjuvant therapy.
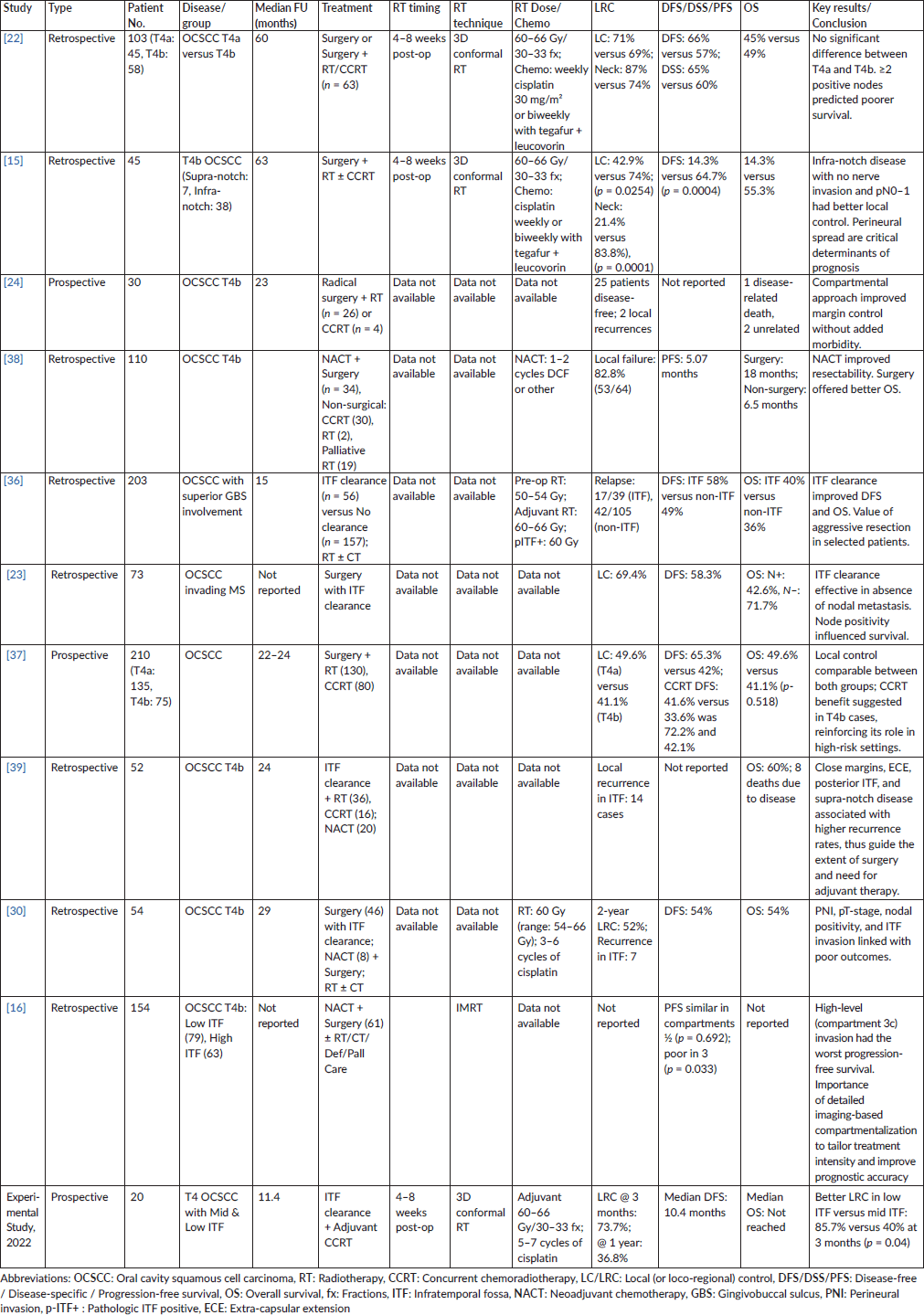
We observed a 3-month LRC rate of 73.7% and 1-year LRC of 36.8%, with a median DFS of 10.4 months. These outcomes are comparable to previously reported prospective and retrospective studies. For example Katna et al [30] Also reported acceptable disease control following compartmental resections with adjuvant therapy. Our findings align with Liao et al) [15], who showed better outcomes in infra-notch T4b OCSCC compared to more extensive disease. Similarly, Manish Mair et al [37] emphasized the benefit of adjuvant CCRT, particularly in improving DFS for advanced cases thus supporting our choice of intensified postoperative therapy as shown in Table 3.
Importantly, patients with low ITF involvement had better short-term outcomes than those with mid ITF involvement (3-month LRC: 85.7% versus 40%, p = 0.04) though this advantage narrowed at 1 year (42.8% versus 40%, p = 0.50) was not statistically significant, indicating that the short-term benefit seen with low ITF involvement does not persist over longer follow-up.
This supports ITF sub-stratification as a useful prognostic marker, as echoed by recent compartment-based approaches like those described by Liao et al [15], who reported better outcomes in infra-notch tumours and recently by Mahajan et al [16]. In cases of T4b oral cavity cancer, the outcomes of CCRT are not very promising; therefore, an initial approach involving ITF clearance followed by adjuvant CCRT may be considered a reasonable treatment option for these patients [35].
Table 3. Showing characteristics and outcomes from comparative studies.
Previous studies have shown that early invasion of the retroantral fat pad indicates posterior spread from buccal or alveolar primaries into the pterygoid muscles. Similarly, involvement of the masseter and medial pterygoid muscles represents involvement into the MS, which is linked to poorer outcomes. Trivedi et al [
Treatment compliance was excellent, and all patients completed planned therapy with integral supportive care strategies such as nutritional supplementation and oral prophylaxis. Grade 3 mucositis and odynophagia were seen in 40% of cases, with no Grade 4 toxicities reported. We have compare outcomes, toxicities were similar to studies by Dewan et al [36] and Mair et al [37], reported acceptable levels of Grade 3 toxicities with no unmanageable adverse effects indicating that CCRT using weekly cisplatin and 3D conformal RT is both feasible and tolerable. Most patients resumed oral intake within 5 weeks, suggesting acceptable functional outcomes post-treatment. The results of such similar studies are tabulated in Table 2.
Our study’s strength lies in its prospective nature, uniform protocol and anatomical stratification, providing focused insight into a rare but operable subgroup of T4b OCSCC. All patients were treated with standardized protocols and timely initiation of RT. Benefit of weekly toxicity monitoring and uniform follow-up added strength to the data quality.
However, certain limitations are the small sample size which limits the generalizability of findings and short follow-up duration is insufficient to draw definitive conclusions about long-term survival or recurrence patterns. Selection bias may be present due to variation in ITF involvement and patient co-morbidities, although strict inclusion criteria were aimed to reduce it. Moreover patients with similar staging who were unfit for surgery or received non-surgical management were not included as part of this prospective study protocol, so prevents our ability to attribute outcomes solely to the intervention. Future prospective studies including all cT4b patients treated during the study period should include comparative arm of surgery alone or non-surgical approaches to provide more generalizable and unbiased conclusions.
Although our study provides important insights into the feasibility and short-term outcomes of surgery with ITF clearance followed by adjuvant CCRT in selected T4b OCSCC patients, these results should be interpreted as early prospective observations. Given the aggressive biology of T4b disease and the potential for late recurrences, these findings must be regarded as preliminary rather than definitive, highlight the importance of extended follow-up and validation in larger multicentre studies, ideally using advanced RT techniques, integrating advanced imaging and biological markers. Optimization of treatment based on ITF compartment involvement may guide to improve result.
Conclusion
In conclusion, this prospective study supports the use of surgery with ITF clearance followed by adjuvant CCRT as a viable treatment approach for selected patients with advanced OCSCC involving the low and mid ITF. The approach is not only feasible but also associated with encouraging early outcomes and manageable toxicity. These results are early observations and not definitive. Because of the aggressive nature of T4b disease and possible late recurrences, longer follow-up and larger studies are needed to validate these findings.
Conflicts of interest
The authors have no conflicts of interest to declare.
Funding
We also did not receive any funding for the present study from any source.
Informed consent
Written informed consent was taken from all patients prior to the enrolment.
Author contributions
Conceptualization: PM, RH, AKG, MR; Investigation and methodology: PM, RH, AKG, MR, RK, SPM, AKS, AB, AS, AA; Supervision: RH, MR, AKG, RK, SPM, AKS, AB; Data curation: PM, MR, AKG, RK, RH, SPM, AKS, AB, AP, VKM, RP, AM, VG, BPV; Analysis and interpretation: RH, MR, AKG, RK, SPM, AKS, AB, AP, VKM, SM; Writing of the original manuscript: PM, RH, AKG, MR; Writing of the review and editing: PM, RH, AKG, MR. All authors have proofread and approved the final version. All authors have contributed significantly to the article. All authors are in agreement with the concept of the manuscript.
Permissions: Approval by Institutional Ethics Committee (IEC No.166/20/Res Cell /RMLIMS/ 2020). The authors acknowledge that the full article has not been published in any journal nor it is under review in any other journal for publication.
Supporting information: Additional supporting information may be found online in the Supporting Information section at the end of the article.
References
1. Bray F, Laversanne M, and Weiderpass E, et al (2024) Global cancer statistics 2022: gLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [Internet] CA Cancer J Clin 74(1) 31–58 [https://acsjournals.onlinelibrary.wiley.com/doi/10.3322/caac.21834] (Accessed 5 Jul 2024)
2. Pathak KA, Gupta S, and Talole S, et al (2005) Advanced squamous cell carcinoma of lower gingivobuccal complex: patterns of spread and failure [Internet] Head Neck 27(7) 597–602 [https://onlinelibrary.wiley.com/doi/10.1002/hed.20195] https://doi.org/10.1002/hed.20195 PMID: 15825204
3. Peters LJ, Goepfert H, and Ang KK, et al (1993) Evaluation of the dose for postoperative radiation therapy of head and neck cancer: first report of a prospective randomized trial Int J Radiat Oncol Biol Phys 26(1) 3–11 https://doi.org/10.1016/0360-3016(93)90167-T PMID: 8482629
4. Ang KK, Trotti A, and Brown BW, et al (2001) Randomized trial addressing risk features and time factors of surgery plus radiotherapy in advanced head and neck cancer Int J Radiat Oncol Biol Phys 51(3) 571–588 https://doi.org/10.1016/S0360-3016(01)01690-X PMID: 11597795
5. Suwinski R, Bankowska-Wozniak M, and Majewski W, et al (2005) Randomized clinical trial on continuous 7-days-a-week postoperative radiotherapy for high-risk squamous cell head and neck cancer: a report on acute normal tissue reactions Radiother Oncol 77(1) 58–64 https://doi.org/10.1016/j.radonc.2005.07.007 PMID: 16157401
6. Bernier J, Domenge C, and Ozsahin M, et al (2004) Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer [Internet] N Engl J Med Internet 350(19) 1945–1952 [https://www.nejm.org/doi/full/10.1056/NEJMoa032641] https://doi.org/10.1056/NEJMoa032641
7. Cooper JS, Pajak TF, and Forastiere AA, et al (2004) Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck N Engl J Med 350(19) 1937–1944 https://doi.org/10.1056/NEJMoa032646 PMID: 15128893
8. Patil VM, Prabhash K, and Noronha V, et al (2014) Neoadjuvant chemotherapy followed by surgery in very locally advanced technically unresectable oral cavity cancers Oral Oncol 50(10) 1000–1004 https://doi.org/10.1016/j.oraloncology.2014.07.015 PMID: 25130412
9. Amin MB, Greene FL, and Edge SB, et al (2017) The eighth edition AJCC cancer staging manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging CA Cancer J Clin 67(2) 93–99 PMID: 28094848
10. Arya S, Rane P, and D’Cruz A (2012) Infratemporal fossa, masticator space and parapharyngeal space: can the radiologist and surgeon speak the same language? Int J Otorhinolaryngol Clin 4(3) 125–131 https://doi.org/10.5005/jp-journals-10003-1098
11. Oliver ER and Gillespie MB (2010) Deep neck space infections Cummings otolaryngology: head and neck surgery 5th (Philadelphia: Mosby Elsevier) p 226
12. Standring S, Ellis H, and Healy J, Johnson D, Williams A, Collins P, et al. Gray’s anatomy: the anatomical basis of clinical practice. 39th ed. Edinburgh: Elsevier Churchill Livingstone; 2005.
13. Standring S (2020) Gray’s anatomy: the anatomical basis of clinical practice [Internet]. 42nd ed. (London: Elsevier) [cited 2024 Jul 6]. Available from: https://shop.elsevier.com/books/grays-anatomy/standring/978-0-7020-7705-0
14. Trivedi N (2015) Tumors of infratemporal fossa [Internet] Atlas of head and neck cancer surgery (New Delhi: Springer India) p 223 [https://link.springer.com/10.1007/978-81-322-2050-3_13] https://doi.org/10.1007/978-81-322-2050-3_13
15. Liao CT, Ng SH, and Chang JTC, et al (2007) T4b oral cavity cancer below the mandibular notch is resectable with a favorable outcome Oral Oncol 43(6) 570–579 https://doi.org/10.1016/j.oraloncology.2006.06.008
16. Mahajan A, Agarwal U, and Ashtekar RM, et al (2025) Compartmentalization of high infratemporal fossa in oral cavity squamous cell carcinomas and its impact on clinical outcome Curr Oncol 32(2) 99 https://doi.org/10.3390/curroncol32020099 PMID: 39996899 PMCID: 11854881
17. Tryhus MR, Smoker WR, and Harnsberger HR (1990) The normal and diseased masticator space [Internet] Semin Ultrasound CT MR 11(6) 476–485 [https://europepmc.org/article/med/2275809]
18. Wei Y, Xiao J, and Zou L (2007) Masticator space: cT and MRI of secondary tumor spread AJR Am J Roentgenol 189(2) 488–497 https://doi.org/10.2214/AJR.07.2212 PMID: 17646477
19. Yousem DM, Gad K, and Tufano RP (2006) Resectability issues with head and neck cancer AJNR Am J Neuroradiol 27(10) 2024–2036 PMID: 17110661 PMCID: 7977212
20. Agarwal JP, Nemade B, and Murthy V, et al (2008) Hypofractionated palliative radiotherapy for advanced head and neck cancer Radiother Oncol 89(1) 51–56 https://doi.org/10.1016/j.radonc.2008.06.007 PMID: 18649963
21. Corry J, Peters LJ, and Costa ID, et al (2005) The “QUAD SHOT”—a phase II study of palliative radiotherapy for incurable head and neck cancer Radiother Oncol 77(2) 137–142 https://doi.org/10.1016/j.radonc.2005.10.008 PMID: 16260054
22. Liao CT, Chang JTC, and Wang HM, et al (2006) Surgical outcome of T4a and resected T4b oral cavity cancer Cancer 107(2) 337–344 https://doi.org/10.1002/cncr.21984 PMID: 16770782
23. Ota Y (2017) Infratemporal surgery for oral cancer invaded to masticator space Int J Oral Maxillofac Surg 46 372–373 https://doi.org/10.1016/j.ijom.2017.02.1253
24. Trivedi NP, Kekatpure V, and Kuriakose MA (2012) Radical (compartment) resection for advanced buccal cancer involving masticator space (T4b): our experience in thirty patients Clin Otolaryngol 37(6) 477–483 https://doi.org/10.1111/j.1749-4486.2012.02529.x PMID: 23253342
25. Trivedi N, Kekatpure V, and Shetkar G, et al (2015) Pathology of advanced buccal mucosa cancer involving masticator space (T4b) Indian J Cancer 52(4) 611–616 https://doi.org/10.4103/0019-509X.178410
26. Bang BA, Arun P, and Sharan R, et al (2014) Infratemporal fossa clearance for oral squamous cancer: is it time to shift the paradigm? J Clin Oncol 32(15) e17047 https://doi.org/10.1200/jco.2014.32.15_suppl.e17047
27. Nanda M and Mohiyuddin A (2015) Neoadjuvant chemotherapy: role in locoregionally advanced oral cancers Int J Med Sci Public Health 4(6) 1–5 https://doi.org/10.5455/ijmsph.2015.20022015146
28. Singh A, Patil VM, and Noronha V, et al (2022) Phase 3 randomized study comparing docetaxel-platinum with docetaxel-platinum-5 fluorouracil as neoadjuvant chemotherapy in technically unresectable oral cancer J Clin Oncol 40(16) 6013 https://doi.org/10.1200/JCO.2022.40.16_suppl.6013
29. Noronha V, Dhanawat A, and Patil VM, et al (2024) Long-term outcomes of neoadjuvant chemotherapy on borderline resectable oral cavity cancers: real-world data of 3266 patients and implications for clinical practice Oral Oncol 148 106633 https://doi.org/10.1016/j.oraloncology.2023.106633
30. Katna R, Kalyani N, and Singh S, et al (2020) Compartmental clearance of infratemporal fossa for T4b carcinoma of buccal mucosa/alveolus: clinical outcomes Indian J Surgical Oncol 11(2) 316–320 https://doi.org/10.1007/s13193-020-01057-y
31. Kurokawa H, Yamashita Y, and Takeda S, et al (2002) Risk factors for late cervical lymph node metastases in patients with stage I or II carcinoma of the tongue Head Neck 24(8) 731–736 https://doi.org/10.1002/hed.10130 PMID: 12203797
32. Evans M and Beasley M (2018) Target delineation for postoperative treatment of head and neck cancer Oral Oncol 86 288–295 https://doi.org/10.1016/j.oraloncology.2018.08.011 PMID: 30409314
33. Lapeyre M, Racadot S, and Renard S, et al (2022) Radiotherapy for oral cavity cancers Cancer Radiother 26(1) 189–198 https://doi.org/10.1016/j.canrad.2021.11.012
34. National Cancer Institute (2017) Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0 (Bethesda: NCI)
35. Mishra VK, Gandhi AK, and Rastogi M, et al (2023) Retrospective analysis of clinical outcome of 100 inoperable oral cavity carcinoma treated with definitive concurrent chemoradiotherapy with or without induction chemotherapy Ecancermedicalscience [Internet] 17 1630 [https://ecancer.org/en/journal/article/1630]
36. Dewan AK, Dabas SK, and Pradhan T, et al (2014) Squamous cell carcinoma of the superior gingivobuccal sulcus: an 11-year institutional experience of 203 cases Jpn J Clin Oncol 44(9) 807–811 https://doi.org/10.1093/jjco/hyu088 PMID: 25009221
37. Mair MD, Sawarkar N, and Nikam S, et al (2018) Impact of radical treatments on survival in locally advanced T4a and T4b buccal mucosa cancers: selected surgically treated T4b cancers have similar control rates as T4a Oral Oncol 82 17–22 https://doi.org/10.1016/j.oraloncology.2018.04.019 PMID: 29909893
38. Prabhash K, Joshi A, and Patil V, et al (2013) Is there a role of induction chemotherapy followed by resection in T4b oral cavity cancers? Indian J Cancer 50(4) 349–353 https://doi.org/10.4103/0019-509X.123627 PMID: 24369216
39. Mohiyuddin SMA, Harsha P, and Maruvala S, et al (2018) Outcome of compartment resection of locally advanced oral cancers extending to infratemporal fossa: a tertiary rural hospital experience Eur Arch Otorhinolaryngol 275(11) 2843–2850 https://doi.org/10.1007/s00405-018-5124-z PMID: 30225567