Genetic association of IL-1B-511 (C/T), TNF-A-308 (G/A) and IL-10-1082 (G/A) with gastric cancer risk in Colombian populations
Kevin Guzmán1, Vanessa Pabón1, Andrés Angulo1, Luis E Bravo2, Arsenio Hidalgo1,3, Carol Rosero4 and Alvaro Pazos1,5
1Grupo Salud Publica, Centro de Estudios en Salud, Universidad de Nariño, Pasto 520001, Colombia
2Registro Poblacional de Cáncer de Cali, Department of Pathology, School of Medicine, Universidad del Valle, Cali 760043, Colombia
3Departamento de Matematicas y Estadistica, Universidad de Nariño, Pasto 520001, Colombia
4Facultad de Medicina, Universidad Cooperativa de Colombia, Pasto 520001, Colombia
5Departamento de Biología, Universidad de Nariño, Pasto 520001, Colombia
Abstract
Background: Gastric cancer (GC) incidence varies markedly across Colombia. Host genetic polymorphisms in cytokine genes modulate the inflammatory response to Helicobacter pylori and may influence disease progression.
Objective: To evaluate IL-1B-511, IL-10-1082 and TNF-A-308 polymorphisms and their association with gastric precursor lesions in Colombian populations with contrasting GC risk.
Methods: We studied 160 dyspeptic patients from Túquerres (high risk) and 105 from Tumaco (low risk). Gastric lesions were classified using the Sydney system. Cytokine polymorphisms were genotyped by polymerase chain reaction–restriction fragment length polymorphism and sequencing. Associations were assessed using logistic regression.
Results: The IL-1B-511TT and IL-10-1082AA genotypes were significantly more frequent in Túquerres than in Tumaco (p < 0.05). Individuals carrying the IL-10-1082AG genotype had a significantly higher risk of gastric atrophy (odds ratio = 5.45; 95% confidence interval, 1.0–30.59).
Conclusion: IL-10-1082AG is associated with increased risk of gastric atrophy in high-risk Colombian populations, supporting its potential role as a biomarker of GC susceptibility.
Keywords: gastric cancer biomarkers, interleukin 1β, interleukin 10, gastric cancer, gastric atrophy, Helicobacter pylori
Correspondence to: Kevin Guzmán
Email: keving346@gmail.com
Published: 07/04/2026
Received: 07/09/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Background
Gastric cancer (GC) is the fifth most frequent malignant neoplasia and the third leading cause of death by cancer worldwide. Also at a global scale, the incidence and mortality rates and the GC risk vary [1, 2]. In Colombia, GC represents the main cause of death by cancer and presents a marked geographical pattern with respect to its incidence distribution, being higher in mountainous regions and lower in coastal areas of the national territory [3, 4].
Helicobarter pylori (H. pylori) infection is the principal etiological factor involved in the development of GC [2, 5–7]; however, the high infection prevalence is not a predictor for the disease incidence [1, 6, 8, 9]. There are other risk factors that are involved with the GC pathogenesis, such as the environmental and hygiene-sanitary factors, diet, host-H. pylori coevolution and human genetic susceptibility [8–13].
The GC human genetic susceptibility is mediated by pro-inflammatory and anti-inflammatory cytokine polymorphisms that participate in the immune response to H. pylori infection. The expression persistence of pro-inflammatory interleukin (IL) polymorphisms is an important factor that contributes to the transformation from a normal gastric mucosa to one displaying precancerous lesions [14–17]. Nevertheless, the evidence that links these polymorphisms with gastric lesions is dependent on the analysed population. For instance, some studies demonstrated a positive association between the genotypes IL-1B-511 and GC risk in Caucasian populations [15, 18, 19], whereas no association of this genotype was found in Asian [20, 21] and Latin American populations from Costa Rica [22], Brazil [23] and Venezuela [24], in contrast to what was reported for some populations in Colombia where an association was found [25, 26]. The genetic susceptibility due to polymorphisms in pro-inflammatory cytokine genes IL-1B-511, FNT-A-308 and anti-inflammatory cytokines IL-1RN and IL-10 are considered an essential factor in the malignant transformation of the normal gastric mucosa. These cytokines participate in the immune response to infections and modulate the type of severity of the gastric inflammation [14, 18]. The opposite results in the analysed associations might indicate that the ethnic differences in the populations influence the distribution of the allelic and genotypic frequencies of these polymorphisms [19, 26, 27].
The Department of Nariño in Colombia represents an important study model to elucidate the GC etiopathogenesis due to its unique geographical, sociocultural and ethnic situation, with an Andean zone of high mountains inhabited by a population of Amerindian origin (67%) and a high GC incidence, which is considered one of the highest in the world (150/100.000 inhabitants). Nariño also has a coastal zone with a population of mostly African origin (58%) with a low GC incidence (6/100.000 inhabitants) [3, 9]. Taking into account the contrasting GC risk in these populations and the contrasting association of cytokine polymorphisms, we investigate what is the relationship of IL-1B-511, IL10-1082 and TNF-A-308 polymorphisms with gastric lesions in two Colombian populations with opposite risk of GC.
Methods
Subjects and samples
We included 265 subjects between the ages of 19–67, with dyspepsia symptoms. Of these, 44.2% (63/160) of men were from Túquerres, a population located in the Andean mountains of Colombia with a high prevalence of H. pylori and preneoplastic lesions. The remaining 41% (38/105) were men from Tumaco, a population located on the Pacific coast with a low GC risk [9]. We analysed 52 patients with GC precursor lesions in these two populations: eight with atrophic chronic gastritis (ACG) and 44 with intestinal metaplasia (IM) and 213 control group patients with non-atrophic gastritis (NAG). We obtained four biopsies from the gastric antrum and gastric body for their histopathological evaluation and H. pylori diagnosis. We obtained 5 mL of total blood for the evaluation of biomarkers of genetic susceptibility to GC.
Posterior to the signature of the informed consent, the participants were given a survey about their sociodemographic factors. This study was approved by the Human Ethics Committee of both Universidad del Valle and Universidad de Nariño.
Histopathology procedures
The gastric lesions were interpreted according to the Sydney classification system [28]. The presence of H. pylori was determined by Giemsa stain. The classified categories were: NAG, ACG and IM. Patients with NAG were defined as a control group and patients with ACG and IM as a group of cases.
DNA extraction
Genomic DNA was extracted from blood using the Ultra Clean Blood Spin Isolation Kit (Mobio, Technologies Inc, San Diego, USA). DNA concentration was determined using NanoDropTM (2000/2000c Spectrophotometers-Thermo Scientific (Thermo Fisher Scientific, Waltham, MA, USA).
IL-1B-511 (C/T) polymorphism
Polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLPs) was used to genotype the IL-1B-511 (C/T) polymorphism. Initially, a 304 bp fragment containing the polymorphic position 511 (C-T transition) was amplified using the primers: 5’-TGGCATTGATCTGGTTCATC-3’ and 5’-GTTTAGGAATCTTCCCACTT-3’ [29, 30], followed by digestion with AvaI restriction enzyme. The PCR conditions were the following, using a 30 µL total volume containing: 1 μL of DNA concentration between 5 and 130 ng/μL; 0.5 nM of each primer; 0.1 nM of the dNTPs mix; 0.5 mM of MgCl2 and 1.25 U of GoTaq DNA Polymerase (Promega®). Amplification conditions: 94°C/5 minutes, followed by 34 amplification cycles (94°C/0.45 second, 57°C/0.45 second and 72°C/0.45 second) and a final extension at 72°C/7 minutes, using a thermocycler (T100TM Biorad). The RFLPs of the PCR products were carried out during 16 h at 37°C using a 30.3 µL final volume containing: 10 µL PCR product, 3 U AvaI restriction enzyme (Eco881) dissolved in 2 µL 10X Tango buffer (Thermo Scientific). The digestion reaction was carried out in the presence of the normal allele (C) and generated two bands of 190 and 114 bp. The characteristic bands for each genotype were: normal homocygous individuals CC (190 and 114 bp), homozygous mutant TT (304 pb) and heterozygous CT (304, 190 and 114 bp) [29, 30]. The restriction fragments from this and the following two digestion reactions were separated by electrophoresis using 1.5% agarose gels stained with 0.4 µL GelRedTM (Biotium, 10.000X) [29].
IL-10-1082 (G/A) polymorphism
We applied PCR-RFLPs to genotype the IL-10-1082 (G/A) polymorphism. We first amplified a 360 bp fragment that contains the polymorphic region -1082 (G-A Transition) using the primers: 5’-CCAAGACAACACTACTAAGGCTTCTTGAGG-3’ and 5’AGGTAGTGCTCAC CATGACC-3’ [29, 30], and the obtained products were subjected to restriction reaction with BseRI. PCR was performed in a final volume of 30 µL containing 2 µL DNA at 5–130 ng/µL; 0.5 nM of each primer; 0.3 nM dNTPs mix; 0.5 mM MgCl2 and 1.5 U of GoTaq DNA Polymerase (Promega®). The amplification step was as follows: 95°C/10 minutes, followed by 35 cycles (95°C/0.30 second, 58°C/0.30 second y 72°C/0.45 second) and a final extension at 72°C/10 minutes using the same thermocycler as above. RFLP digestions were carried out for 45 minutes at 37°C using a 15 µL final volume, containing: 5 µL PCR product, 2 U BseRI diluted in 1.5 µL 10X NE buffer (New England, BioLabbs®). The digestion was produced in the presence of the normal allele (G) and the produced bands for each genotype were: GG normal homozygous individuals (320 bp), AA mutant homozygous (360 bp) and GA heterozygous (360, 320 bp) [29, 30].
TNF-A-308 (G/A) polymorphism
PCR-RFLP was used to genotype the TNF-A-308 (G/A) polymorphism. We initially amplified a 107 bp fragment that includes the polymorphic region -308 (G-A Transition), using the primers: 5’-AGGCAATAGGTTTTGAGGGCCAT-3’ and 5’-TCCTCCCTGCTCCGATTCCG-3’ [29, 30], followed by digestion with Nco1 restriction enzyme. The PCR reaction mix was similar to the one used for IL-1B gene. The amplification steps included: 94°C/5 minutes, 30 cycles (94°C/1 minute, 57°C/1 minute and 72°C/1 minute) and final extension at 72°C/5 minutes in the same thermocycler. The RFLPs of the PCR products were carried out for 16 h at 37°C in a final volume of 30.3 µL that contained 10 µL PCR, 3 U Nco1 enzyme diluted in 2 µL 10X Tango buffer. The digestion was developed in the presence of the normal allele (GG) that generates a 87 bp band; the heterozygous (GA) produce two bands of 87 and 107 bp, and the mutant homozygous (AA) generates a 107 bp band [29, 30].
Statistical analysis
We evaluated the adjustment of the Hardy–Weinberg equilibrium (EH-W) using the allelic and genotypic frequencies of the control group from the two studied populations through the Markov Chain approach with 100,000 permutations in the Arlequin software version 3.5.2. We made contingency tables based on the χ2 test in order to determine whether there are significant differences in the genotypic and allelic distributions between the two analysed zones and to determine their relationship with the type of gastric lesion. The magnitude of the association between the genotypes and the gastric lesion was expressed as odds ratio (OR) with a 95% confidence interval (95% CI) that was estimated via binary logistic regression adjusted by sex, age, H. pylori infection and polymorphisms simultaneously. An association was considered significant if it showed a p-value ≤ 0.05. The statistical analyses were carried out using the SPSS program version 22.0.
Results
The 52 patients with GC precursor lesions were classified as GCA [8] and IM [44], and 213 patients were placed in the control group NAG. The infection prevalence of H. pylori in Tumaco was 94.3%, which is significantly higher than the one in Tuquerres (85.6%) (p < 0.05). We observed that the type of gastric lesion is dependent on the geographical origin, with a borderline significance (p = 0.07). Here, the IM lesion was the more frequent in the high-risk population (68.2%) than in the low-risk population (31.8%), whereas NAG showed a higher frequency in Tumaco (75%) than in Túquerres (25%). We combined the ACG and IM cases in a single group (ACG) due to the low frequency of the number of GC precursor lesions and to carry out the statistical analysis. With respect to the sociodemographic characteristics of the patients, we found a significant difference (p < 0.05) between the cases and control groups for both populations in reference to sex. We observed that gastric atrophy was more frequent in male (30.7%) than in females (12.8%), and men have a three times higher risk of developing ACG compared to women, independently of their origin. A similar trend was observed with age, where the GC precursor lesions are more frequent in individuals older than 30 years of age (22.2%) compared to younger individuals (5%) (p = 0.01) (Table 1). In addition, individuals older than 30 years of age had a 5.4 higher risk of developing gastric atrophy compared to younger people. Finally, we did not find significant differences between H. pylori infection and the type of gastric lesion in individuals from the two studied populations (Table 1).
IL1B-511, IL-10-1082 and TNF-A-308 polymorphisms
The PCR-based amplification of the polymorphic regions of IL-1B, IL-10 and TNF-A genes revealed the presence of the products with the expected polymorphic fragments according to the used primers (Figure 1). The categorisation and classification of the products from PCR-RFLPs allowed us to detect the presence of the three genotypes from each of the polymorphisms in both populations (Figure 2).
Frequencies of cytokine polymorphisms according to population at risk for GC
We analysed 259, 262 and 264 polymorphic profiles from IL-1B-511, IL-10-1082 and TNF-A-308, respectively. It was determined that the group controls from both populations are in EH-W equilibrium for the TNF-A-308 gene polymorphism (p = 0.37 for Tumaco and p = 1.00 for Túquerres). The IL-10-1082 was adjusted to the EH-W equilibrium (p = 0.78) only in the control group from the high-risk population, which means that it is not under the influence of evolutive factors or forces that can modify it. With respect to IL-1B-511 gene polymorphism, the EH-W was not adjusted in either of the two populations (p = 0.001 for Tumaco and p = 0.04 for Túquerres) [26].
Table 1. Distribution of the association of sociodemographic characteristics and H. pylori infection with the type of gastric lesion.
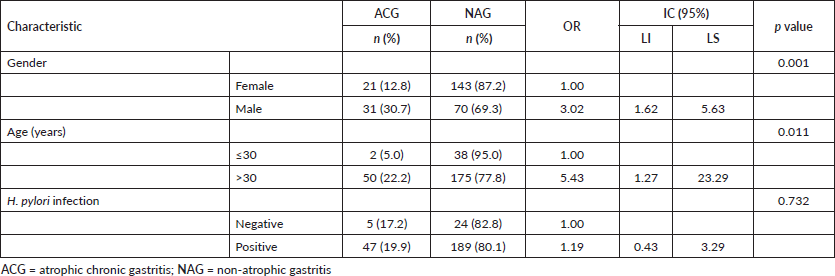
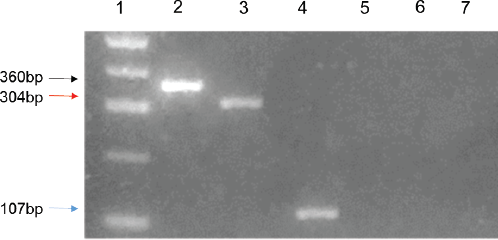
Figure 1. Electrophoretic pattern of the PCR products from the polymorphic regions -1082, -511 and -308 in IL10, IL1-B and TNF-A genes, respectively. 1.5% agarose gel electrophoresis. Lane 1: molecular marker (100 bp DNA ladder); lane 2: the black arrow indicates the 360 bp band that corresponds to the amplification product of -1082 region of IL-10 gene; lane 3: the red arrow indicates the 304 bp band corresponding to the amplification product or the -511 region of the IL1-B gene; lane 4: the blue arrow marks the 107 bp band that represents the amplification product of the -308 region of the gene TNF-A; lanes 5–7: negative controls for each gene.
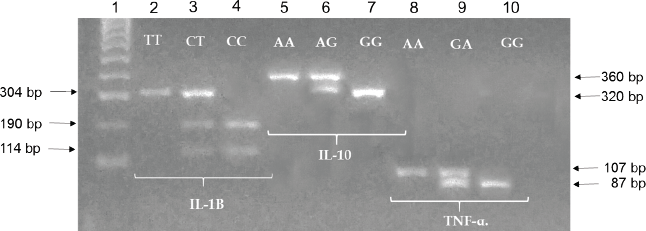
Figure 2. Electrophoretic pattern of the digestion products by RFLP of the polymorphic regions -511, -1082 and -308, IL1-B, IL-10 and TNF-A genes, respectively. 1.5% agarose gel electrophoresis. Lane 1: molecular marker (100 bp DNA ladder); lanes 2–4: the arrows indicate the 304, 190 and 114 bp bands of the digestion products obtained with AvaI enzyme, corresponding to the mutant homozygous genotypes (TT), heterozygous (CT) and homozygous (CC) of the -511 region of IL1-B gene. Lanes 5–7: the arrows indicate the 360 and 320 bp bands produced by digestion with BseRI restriction enzyme, which correspond to homozygous genotype (AA), heterozygous (AA) and homozygous (GG), -1082 region of IL-10 gene. Lanes 8–10: the arrows indicate the 107 and 87 bp band products of the digestion with NcoI that correspond to the AA mutant, GA heterozygous and GG homozygous normal genotypes, -308 region of the TNF-A gene.
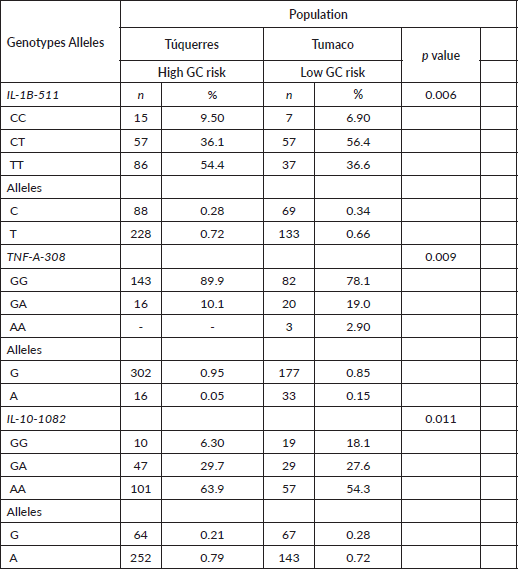
The IL1B-511TT and IL-10-1082AA genotypes, which are carriers of the T and A risk alleles, were significantly more frequent in the GC high-risk population: Túquerres (54.4% and 63.9%) than in the low-risk population: Tumaco (36.6% and 54.3%), respectively, p ≤ 0.05. The distribution of the alleles showed that the patients with high GC risk had a significantly higher frequency of the IL1B-511T mutant alleles (72%) and IL-10-1082A (79%) than the frequencies found for these alleles in the low GC risk patients: Tumaco, p ≤ 0.05 (Table 2). We found that the TNF-A-308AA genotype was absent in the Túquerres population, while in the Tumaco population this genotype was only present in 2.9% of the patients. Similarly, the distribution of the A allele frequency of this genotype is almost null in the two populations (Table 2).
Table 2. Genotypic and allelic frequencies of cytokine polymorphisms according to GC risk population.
Frequencies of cytokine polymorphisms according to gastric lesion type
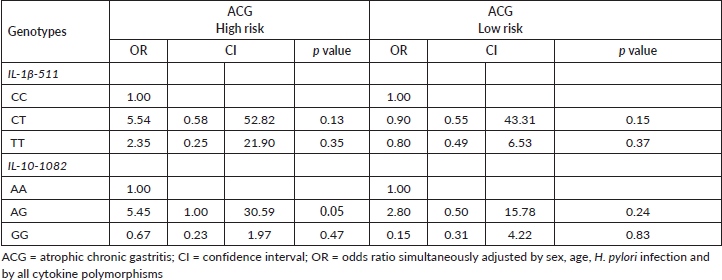
The distribution of the different genotypes within the control group NAG and within the study group ACG showed that the IL1B-511, IL-10-1082 and TNF-A-308 gene polymorphisms had no differences between the type of gastric lesion and the genotypes of (IL) in the two studied populations (Table 3). Independent of population, we found that the most predominant genotype in the ACG patient group was the IL-1B-511CT heterozygous which displayed higher frequency in Túquerres (51.6%) as well as in Tumaco (50%). In the case of the IL-10-1082AA genotype, carriers of the A risk allele were the most prevalent among the ACG patients in both populations, Túquerres (65.6%) and Tumaco (45%). Significant differences were not found in the total sum of frequencies of the risk genotypes versus the normal genotypes in the three studied genes and the type of gastric lesion (Table 3). It was found that the TNF-A-308AA genotype, carrier of the rare A allele, was absent in patients in ACG case groups from both populations, while the TNF-A-308GG genotype, carrier of the frequent allele, was more common in general. Since this polymorphism was absent in the case groups, it was not included in the logistic regression model (Table 3). The results of the analysis of the binary logistic regression which were adjusted simultaneously by sex, age, H. pylori infection and all of the polymorphisms, showed that people that inhabit the high risk zone and are carriers of the heterozygous IL-10-1082AG genotype presented a 5.45 higher probability of developing ACG in comparison to those who were carriers of the IL-10-1082AA genotype (OR = 5.4; 95% CI, 1.0–30.59). With respect to the remaining genotypes, no positive association with gastric lesion was found (Table 4).
Table 3. Genotypic frequencies of cytokine polymorphisms according to the lesion and GC risk population.
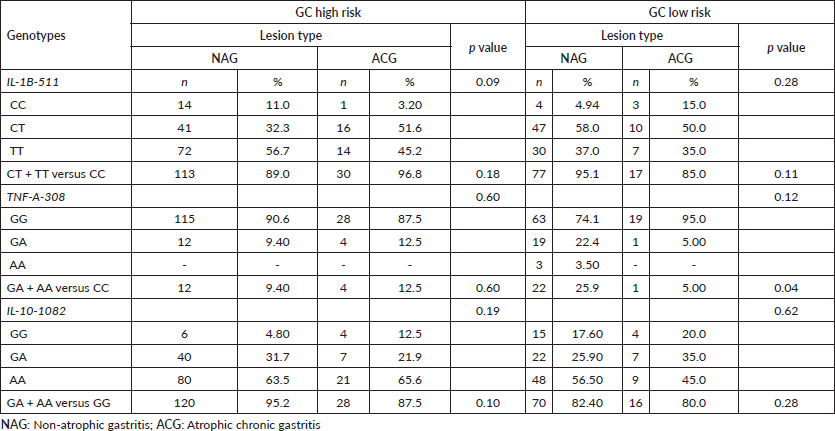
Tabla 4. Risk of developing gastric atrophy according to cytokine genotypes in populations with contrast in the risk of GC.
The results of the allelic and genotypic variants evaluated by PCR-RFLP for the three cytokine genes were concordant with those found by the method of sequencing and identification of mutations. The kappa coefficient κ = 0.83, 0.77 and 0.81 shows that there was a good level of concordance between the results of the allelic and genotypic variants evaluated by PCR-RFLP and those detected by the sequencing method of the IL-1B-511; IL-10-1082 and TNF-A-308 polymorphisms, respectively.
Discussion
The prevalence of H. pylori infection was greater in the low-risk GC population (Tumaco, 94.3%) compared to the high-risk population (Túquerres, 85.65%) [31, 32], but these differences are not significant. However, differences in GC incidence rates exist between these two populations, despite being in close geographic proximity (within 200 km of each other); the incidence is 25 times higher in the Túquerres population, which is located in the Colombian Andes with predominantly Amerindian and European descent, than in the Tumaco population, which is located in the Pacific coast and has a predominantly African ancestry [3, 4, 9]. This phenomenon is similar to the ‘African enigma’, which describes that the H. pylori infection is very high in Africa but GC is less frequent [10]; which is similar to what was observed in the Tumaco population, a phenomenon previously reported as the ‘Colombian enigma’ [4]. Accordingly, in the present study and with relation to the severity of gastric lesions, in the high GC population: Túquerres has a higher prevalence of IM (68.2%) than Tumaco (31.8%), which is considered a premalignant histological lesion in the progression to intestinal gastric adenocarcinoma, described in the GC precancerous cascade model [5, 33, 34]. The risk of developing GC is 90 times higher when IM appears compared to individuals with normal mucosa [34]. These observations suggest that there are other complementary factors of sociocultural type such as diet and the environment [4, 35], as well as bacterial infection, genetic factors associated to humans and biological factors such a sex and age, which are important for the development and progression of the disease [5, 8, 10, 12, 13, 27, 33, 34].
In this work, we found that men have a three times higher risk of developing ACG than women, which is in agreement with another study that reported a two times higher risk of gastric atrophy in men than in women, and that the disease appears at a younger age in males (<55 years old) than in females [34]. In our study, we observed that the ACG risk increases by five times at ages older than 30 in both studied populations, which is in accordance with previous research carried out with Colombian populations that reported a positive association between atrophic gastritis and IM with patients age (40 and 64 years old) [36]. These findings must motivate people to practice gastroscopy as reported by Haruma et al [37], who found that atrophic gastritis and IM significantly increase the GC risk in young Asian patients at ages of 18–29.
According to several reports, genetic polymorphisms that encode for different anti- and pro-inflammatory cytokines are considered a key factor in the process of gastric carcinogenesis. Thus, their study can be used as a tool in the search for variations in the individual genetic susceptibility to GC [5, 8, 12, 15, 16, 27].
We found significant differences in the distribution of frequencies of the IL-1B-511TT and IL-10-1082AA genotypes between the two populations, being more frequent in the patients from the high GC risk zone (54% and 63.9%, respectively) in comparison to patients from the low risk zone (36.6% and 54.3%, respectively). Similar results were observed by Zabaleta et al [43], in which the allelic and genotypic frequencies of IL-1B-511TT and IL10-1082AA were higher in individuals of high GC risk, Afroamericans (26.8% and 41.2%, respectively) than in Caucasians of low risk (12.1% and 26%, respectively). However, the distribution of frequencies of these genotypes was more elevated in the Colombian populations found in our study. Here, the allelic frequencies of the risk genotypes IL-1B-511T and IL-10-1082A were higher in patients of GC high risk: Túquerres (72% and 79%, respectively) was higher than in the low risk population of Tumaco (66% and 72%).
The differences in the distribution of the allelic and genotypic frequencies of the IL-1B-511 and IL-10-1082 polymorphisms between the two studied populations are influenced possibly by the genetic composition of the ethnic groups from these regions [20, 40, 47–49]. The high frequency of the IL-1B-511T high risk allele in the population of Túquerres: high risk population, was similar to the one reported for Latin American populations such as Perú (79%), Venezuela (52%) and Ecuador (62.9%) [24, 38, 44], meanwhile the frequency of the IL-10-1082A low risk allele found in the same population was higher and similar to the one reported for Asian and Latin populations [25, 40, 41, 47, 48, 50]. The IL-1B-511TT and IL-10-1082AA genotypes are associated with a higher risk for GC precursor lesions [24, 45], in Caucasian populations [15, 18], Colombia [25, 40], Venezuela [42], USA [14], Asian and Japan [46]. The previous studies are supported by cumulative evidence showing that IL-1β induced by H. pylori is a pro-inflammatory cytokine that modulates the biological function of various types of gastric epithelial cells [14], is an in vivo inhibitor of gastric acid that initiates and amplifies the inflammatory response and induces hypochlorhydria and gastric atrophy [14, 15, 33]. This cytokine is increased in carriers of the genotypes of the IL-1B-511, IL-1B-31 and IL-1RNA2*A2 genotypes and, in thus, this increases the risk of precancerous lesions and adenocarcinomas of distal type [30, 34, 35].
A positive association was found between the IL-1B-511 polymorphism and ACG in the high risk population. The carrier patients of the IL-1B-511CT and IL-1B-511TT genotypes displayed a 5.54 and 2.35 higher risk increase of ACG (OR = 5.54; 95% CI, 0.58–52.82); (OR = 2.35; 95% CI, 0.25–21.9). Similar results in Colombian patients have been reported [25], where an association between the IL-1B-511TT genotype and IM was described (OR = 4.05; 95% CI, 1.35–12.10). A meta analysis by Peleteiro et al [41] also reported a positive association between the IL-1B-511CT genotype with IM in a high GC risk population of Caucasian origin, but no association in a population of low GC risk of African origin [37]. The biological effect of the IL-1β in the risk of gastric carcionogenesis continues to show disparities as shown in various meta analyses of association [26, 38, 39] and no association [40].
In this study, we found that the TNF-A-308 AA genotype was absent in the population with high GC risk, while in the low risk population it is only present in 2.9% of patients from the NAG control group. The allelic and genotypic frequencies for this polymorphism follow the global tendency that exhibits a low frequency of the A mutant allele and a significant increase of the G normal allele [38]. Thus, this polymorphism is not associated with GC precursor lesions in neither of the two studied populations, which is in accordance with the findings reported by Acosta et al [38], Torres et al [39], Martínez et al [40], who worked with Peruvian and Colombian populations. Similar results showing no association were described in the meta analysis of Peleteiro et al [41] and in the study by Kato et al [42], in Venezuela. As demonstrated by both the literature and this current study, it can be suggested that the TNF-A-308 polymorphism should not be considered as a predictor for GC risk in Colombian populations. That the TNF-A-308 polymorphism is not a relevant predictor factor for GC risk in Hispanic populations is a view that is shared by other authors [38].
The in vivo function of the pro-inflammatory cytokine IL-10 is to limit inflammatory responses [16, 17, 29]. The IL-10-1082GG genotype is associated with both an elevated production of IL-10 and inhibitory effects over the inflammatory function of other cytokines like IL-1β y TNF-A. In contrast, the IL-10-1082AA genotype is associated with both low IL-10 concentrations in gastric mucosa and GC [47]. We found that the G allele present in the IL-10-1082GG+GA versus GG genotypes is significantly associated with ACG in control group patients from the population of Túquerres: high GC risk. This association has not been reported for populations from Tumaco (low GC risk). Previous reports have shown that IL-10-1082AA and IL-10-1082AG genotypes are associated with GC in Caucasian and Asian populations [13, 26, 27], respectively; the latter having G as the risk allele, which is similar to our observations for high risk populations. We did not identify an association between the IL-10-1082AA and the risk of developing GCA, a result that is consistent with previous studies that did not find significant differences in the distribution of IL-10-1082 AA+GA versus GG genotypes between control and GCA groups (OR = 2.91; 95% CI, 0.76–11.08) [25]. In this respect, our results clearly indicate that individuals from the GC high risk population in Colombia: Túquerres and carriers of the IL-10-1082AG mutant genotype have a 5.45 higher risk of developing gastric atrophy in comparison to those carrying the IL-10-1082AA genotype (OR = 5.45; 95% CI, 1–30.59) within the same studied population. It is important to mention that this association was not observed in individuals that inhabit populations with low GC risk: Tumaco. Similarly, Loh et al [20] has reported that in Asian populations, which are categorised as high GC risk, this genotype is associated with GC (OR = 1.56; 95% CI, 1.23–1.96). Zhou et al [50] have reported comparable results (OR = 1.50; 95% CI, 1.06–2.11). This genotype is associated with a low IL-10 cytokine expression in gastric mucosa, reduction of its anti-inflammatory effect and subsequent loss of inhibition of the pro-inflammatory effect provided by cytokines such as IL-β and TNF-A [47] and induced by H. pylori infection.
Several authors have proposed that the variabilities in the association of the polymorphisms with precancerous lesions and GC in populations around the world are the consequences of differences in the genetic composition of ethnic groups, which affect their frequencies [9, 10, 40, 41, 43, 48]. Our results are in accordance with this idea since the distribution of the genotypic and allelic risk polymorphisms are significantly different between the two studied populations, which displays contrasting features with respect to the ethnic origin (being mostly African in Tumaco and mostly Amerindian in Túquerres). Also in our study, we show strong evidence supporting an association between the IL-10-1082AG mutant phenotype and ACG. It is possible that the previously described evidence in combination with the coevolutionary theory demonstrated in our study (disruption of the host-H. pylori coevolution associated with more severe gastric lesions) [9] determine biological relationships that are less favorable for the host and explain partially the differences between the risk and incidence of GC between the two Colombian populations analysed in this work.
A limitation of this study is the relatively small sample size of some subgroups, particularly the Tumaco population and the patients with gastric atrophy. Although statistically significant associations were detected, a larger sample size would increase statistical power and might reveal additional genotype–phenotype relationships.
The identification of IL-10-1082AG as a genetic risk factor for gastric atrophy in high-risk Colombian populations suggests potential clinical applications. This polymorphism could be incorporated into future risk-stratification models to identify individuals who may benefit from closer endoscopic surveillance or early H. pylori eradication. In high-incidence regions such as Túquerres, genetic screening combined with histological and microbial markers could improve targeted prevention strategies and reduce progression toward GC.
Conclusion
In conclusion, the disparity of the GC risk in the two populations can be the result of (i) the significant differences in the distribution of the allelic and genotypic differences of IL-1B-511 and IL-10-1082 polymorphisms and (ii) the positive association between the IL-10-1082AG genotype with ACG in the high GC risk population in Colombia. Nevertheless, future studies that include the genetic ancestry and the disruption of the H. pylori-host coevolution are needed to improve the understanding of the role of the cytokine polymorphisms in the gastric carcinogenesis.
Acknowledgments
Thanks to biologists Stephany Rosero and Javier Chaleal for their contributions to the technical development of the project: Biomarkers for genetic susceptibility to gastric cancer in Nariño.
Conflicts of interest
The authors declare that they have no competing interests.
Funding
The Molecular Microbiology Lab and Public Health Research Group from Universidad de Nariño supported this investigation. The Universidad del Valle and Universidad Cooperativa de Colombia provided financial support for the co-execution of this research project. Our RC No 693-2014 research grant was financed by the National Program CTel in Health of MINCIENCIAS-Colombia.
Author contributions
All the authors that were involved in the acquisition and interpretation of the results, read and approved the final manuscript; Guzman K, Pazos AJ, Bravo LE and Pabon V designed the research; Pazos A, Pabon V, Rosero C and Angulo A conducted the microbiological and molecular tests; Pazos A, Pabon V, Angulo A, Guzman K and Hidalgo A, analysed the data; Pazos A, Pabon V, Hidalgo A, Angulo A, Rosero C, Guzman K and Bravo LE wrote, edited and revised the manuscript.
References
1. Malfertheiner P, Camargo MC, and El-Omar E, et al (2023) Helicobacter pylori infection Nat Rev Dis Primers 9 19 https://doi.org/10.1038/s41572-023-00431-8 PMID: 37081005 PMCID: 11558793
2. Piazuelo MB and Correa P (2013) Cáncer gástrico: punto de vista Colomb Med 44(3) 192–201 https://doi.org/10.25100/cm.v44i3.1263
3. Guzman KA and Pazos A (2023) Helicobacter pylori: microorganismo patógeno o mutualista en poblaciones colombianas Universidad Y Salud 25(1) 1–6 https://doi.org/10.22267/rus.232501.292
4. Correa P (2013) Gastric cancer: overview Gastroenterol Clin North Am 42(2) 211–217 https://doi.org/10.1016/j.gtc.2013.01.002 PMID: 23639637 PMCID: 3995345
5. Guzman K, Montenegro L, and Pazos A (2023) The Helicobacter pylori single nucleotide polymorphisms (SNPs) associated with multiple therapy resistance in Colombia Front Microbiol 14 1198325 https://doi.org/10.3389/fmicb.2023.1198325
6. Guzman KA, Hidalgo A, and Pazos AJ (2024) Point mutations in furazolidone and rifampicin resistance genes in Helicobacter pylori strains from Colombia Antibiotics 13(7) 643 https://doi.org/10.3390/antibiotics13070643 PMID: 39061325 PMCID: 11274218
7. International Agency for Research on Cancer (1994) IARC working group on the evaluation of carcinogenic risks to humans: schistosomes, liver flukes and Helicobacter pylori IARC. Monogr Eval Carcinog Risks Hum 61 1–241
8. Guzman KA, Daza AP, and Gomez RV, et al (2024) Whole-genome sequences of Helicobacter pylori isolated from patients with high risk of gastric cancer in the Andes of Nariño, Colombia Microbiol Resour Announc 13 https://doi.org/10.1128/mra.01232-23
9. Kodaman N, Pazos A, and Schneider BG, et al (2014) Human and Helicobacter pylori coevolution shapes the risk of gastric disease Proc Natl Acad Sci U S A 1455 111–1460 https://doi.org/ 10.1073/pnas.1318093111
10. Holcombe C (1992) Helicobacter pylori: the African enigma Gut 33 429–431 https://doi.org/10.1136/gut.33.4.429 PMID: 1582581 PMCID: 1374052
11. Burucoa C and Axon A (2004) Epidemiology of Helicobacter pylori infection Helicobacter 9(1) 1–6 https://doi.org/10.1111/j.1083-4389.2004.00248.x
12. Bonequi P, Meneses-González F, and Correa P, et al (2013) Risk factors for gastric cancer in Latin America: a meta-analysis Cancer Causes Control 24 217–231 https://doi.org/10.1007/s10552-012-0110-z
13. Tsuji S (2006) Helicobacter pylori eradication to prevent gastric cancer: underlying molecular and cellular mechanisms World J Gastroenterol 12(11) 1671–1680 https://doi.org/10.3748/wjg.v12.i11.1671 PMID: 16586533 PMCID: 4124339
14. El-Omar EM, Carrington M, and Chow WH, et al (2000) Interleukin-1 polymorphisms associated with increased risk of gastric cancer Nature 404 398–402 https://doi.org/10.1038/35006081 PMID: 10746728
15. Figueiredo C, Machado JC, and Pharoah P, et al (2002) Helicobacter pylori and interleukin genotyping: an opportunity to identify high-risk individuals for gastric carcinoma J Natl Cancer Inst 94(22) 1680–1687 https://doi.org/10.1093/jnci/94.22.1680 PMID: 12441323
16. Ma J, Wu D, and Hu X, et al (2017) Associations between cytokine gene polymorphisms and susceptibility to Helicobacter pylori infection and related gastric cancer, peptic ulcer disease: a meta-analysis PLoS One 12(0176463) e0176463 https://doi.org/10.1371/journal.pone.0176463
17. Moore KW, De Waal Malefyt R, and Coffman RL, et al (2001) Interleukin-10 and the interleukin-10 receptor Annu Rev Immunol 19 683–765 https://doi.org/10.1146/annurev.immunol.19.1.683 PMID: 11244051
18. Rad R (2004) Cytokine gene polymorphisms influence mucosal cytokine expression, gastric inflammation, and host specific colonisation during Helicobacter pylori infection Gut 53 1082–1089 https://doi.org/10.1136/gut.2003.029736 PMID: 15247172 PMCID: 1774164
19. Camargo MC, Mera R, and Correa P, et al (2006) Interleukin-1beta and interleukin-1 receptor antagonist gene polymorphisms and gastric cancer: a meta-analysis Cancer Epidemiol Biomark Prev 15 1674–1687 https://doi.org/10.1158/1055-9965.EPI-06-0189
20. Loh M, Koh KX, and Yeo BH, et al (2009) Meta-analysis of genetic polymorphisms and gastric cancer risk: variability in associations according to race Eur J Cancer 45 2562–2568 https://doi.org/10.1016/j.ejca.2009.03.017 PMID: 19375306
21. Xue H, Lin B, and Ni P, et al (2010) Interleukin-1B and interleukin-1 RN polymorphisms and gastric carcinoma risk: a meta-analysis J Gastroenterol Hepatol 25 1604–1617 https://doi.org/10.1111/j.1440-1746.2010.06428.x PMID: 20880168
22. Alpízar-Alpízar W, Pérez-Pérez GI, and Une C, et al (2005) Association of interleukin-1B and interleukin-1RN polymorphisms with gastric cancer in a high-risk population of Costa Rica Clin Exp Med 5 169–176 https://doi.org/10.1007/s10238-005-0082-3 PMID: 16362796
23. Melo HP, Martins LC, and Dos Santos SE, et al (2009) Interleukin-1B and TNF-α polymorphisms and Helicobacter pylori in a Brazilian Amazon population World J Gastroenterol 15 1465–1471 https://doi.org/10.3748/wjg.15.1465
24. Cañas M, Morán Y, and Rivero MB, et al (2009) Interleukin-1 genetic polymorphism: association with gastric cancer in the high-risk Central-Western population of Venezuela Rev Med Chil 137 63–70 https://doi.org/10.4067/S0034-98872009000100009
25. Martínez T, Hernández GA, and Bravo MM, et al (2014) Pre-cancerous gastric lesions in Colombian patients: association with IL-1B-511, IL-1RN, IL-10-919, IL-10-1082, TNF-α-308 genes polymorphisms, and anti-Helicobacter pylori cagA IgG antibodies Rev Colomb Cancerol 18(8) 8–17 https://doi.org/10.1016/S0123-9015(14)70220-3
26. Martínez T, Hernández-Suárez G, and Mercedes Bravo M, et al (2011) Asociación de los polimorfismos IL-1B-511 e IL-1RN, y Helicobacter pylori cagA positivo con cáncer gástrico en una zona de riesgo alto en Colombia Rev Med Chil 139 1313–1321 https://doi.org/10.4067/S0034-98872011001000010
27. Persson C, Canedo P, and Machado JC, et al (2011) Polymorphisms in inflammatory response genes and their association with gastric cancer: a HuGE systematic review and meta-analyses Am J Epidemiology 173 259–270 https://doi.org/10.1093/aje/kwq370
28. Dixon MF, Genta RM, and Yardley JH, et al (1996) Classification and grading of gastritis: the updated Sydney system Am J Surg Pathol 20 1161–1181 https://doi.org/10.1097/00000478-199610000-00001 PMID: 8827022
29. Rosero-Rojas SC, Chaleal-Cultid JA, and Pazos-Moncayo AJ, et al (2020) Polimorfismos IL1B-511 y TNF-A-308 en una población infectada con Helicobacter pylori de una zona de bajo riesgo de cáncer gástrico en Nariño-Colombia Infectio 24(2) 81–87 https://doi.org/10.22354/in.v24i2.837
30. Perri F, Piepoli A, and Bonvicini C, et al (2005) Cytokine gene polymorphisms in gastric cancer patients from two Italian areas at high and low cancer prevalence Cytokine 30 293–302 https://doi.org/10.1016/j.cyto.2005.01.011 PMID: 15927855
31. Matta AJ, Pazos AJ, and Bustamante-Rengifo JA, et al (2017) Genomic variability of Helicobacter pylori isolates of gastric regions from two Colombian populations World J Gastroenterol 23 800–809 https://doi.org/10.3748/wjg.v23.i5.800 PMID: 28223724 PMCID: 5296196
32. Pazos A (2012) Ancestros filogeográficos, susceptibilidad antimicrobial y marcadores de virulencia de Helicobacter pylori en pacientes con gastritis crónica (Universidad del Valle) [http://bibliotecadigital.univalle.edu.co/bitstream/10893/7793/1/CB-0516176.pdf]
33. Correa P, Haenszel W, and Cuello C, et al (1975) A model for gastric cancer epidemiology Lancet 2 58–60 https://doi.org/10.1016/S0140-6736(75)90498-5 PMID: 49653
34. Sipponen P and Marshall BJ (2000) Gastritis and gastric cancer in Western countries Gastroenterol Clin N Am 29 579–592 https://doi.org/10.1016/S0889-8553(05)70131-X
35. Suárez R, Wiesner C, and González C, et al (2004) Antropología del cáncer e investigación aplicada en salud pública Rev Estud Soc 17 42–55 https://doi.org/10.7440/res17.2004.04
36. Martínez J, Henao S, and Granados C (2007) La gastritis crónica atrófica corporal y la edad Rev Colomb Gastroenterol 22 17–22 [http://www.redalyc.org/html/3377/337729274005/]
37. Haruma K, Komoto T, and Kamada T, et al (2000) Helicobacter pylori infection is a major risk factor for gastric cancer in young patients Scand J Gastroenterol 35 255–259 https://doi.org/10.1080/003655200750024100 PMID: 10766317
38. Acosta O, Solano L, and Huerta D, et al (2012) Variabilidad genética de la respuesta inflamatoria: polimorfismo -511 C/T en el gen IL1β en diferentes subpoblaciones peruanas Fac Med 73 221–225 [http://www.scielo.org.pe/pdf/afm/v73n3/a09v73n3.pdf] https://doi.org/10.15381/anales.v73i3.868
39. Torres M, Acosta C, and Sicard D, et al (2004) Genetic susceptibility and risk of gastric cancer in a human population of Cauca, Colombia Biomédica 24 153–162 https://doi.org/10.7705/biomedica.v24i2.1261
40. Martínez T, Hernández G, and Bravo MM, et al (2011) Polimorfismos genéticos de interleucinas IL-B-511, IL-1RN, IL-10, TNF-α-308 e infección por Helicobacter pylori CagA positivo en cáncer gástrico y úlcera duodenal en diferentes poblaciones en Colombia Rev Colomb Cancerol 15 85–97 https://doi.org/10.1016/S0123-9015(11)70071-3
41. Peleteiro B, Lunet N, and Carrilho C, et al (2010) Association between cytokine gene polymorphisms and gastric precancerous lesions: systematic review and meta-analysis Cancer Epidemiology Biomarkers & Prevention 19 762–776 https://doi.org/10.1158/1055-9965.EPI-09-0917
42. Kato I, Van Doorn LJ, and Canzian F, et al (2006) Host bacterial interaction in the development of gastric precancerous lesions in a high-risk population for gastric cancer in Venezuela Int J Cancer 119 1666–1671 https://doi.org/10.1002/ijc.21979 PMID: 16671087
43. Zabaleta J, Schneider B, and Ryckman K, et al (2008) Ethnic differences in cytokine gene polymorphisms: potential implications for cancer development Cancer Immunol Immunother 57 107–114 https://doi.org/10.1007/s00262-007-0358-4
44. Cumbal N (2010) Estudio de los polimorfismos en las regiones -31(T>C), -511(C>T) y +3954(C>T) del gen Il-1β mediante PCR-RFLP en población ecuatoriana con cáncer gástrico y presencia de Helicobacter pylori [tesis de pregrado en Internet] (Ecuador: Escuela Politécnica del Ejército) [http://www.bibliotecasdelecuador.com/Record/ir-:21000-889]
45. Lundin BS, Enarsson K, and Kindlund B, et al (2007) The local and systemic T-cell response to Helicobacter pylori in gastric cancer patients is characterised by production of interleukin-10 Clin Immunol 125 205–213 https://doi.org/10.1016/j.clim.2007.07.011 PMID: 17826353
46. Furuta T, El–Omar EM, and Xiao F, et al (2002) Interleukin-1β polymorphisms increase risk of hypochlorhydria and atrophic gastritis and reduce risk of duodenal ulcer recurrence in Japan Gastroenterology 123 92–105 https://doi.org/10.1053/gast.2002.34156 PMID: 12105837
47. Won HH, Kim JW, and Kim MJ, et al (2010) Interleukin-10 polymorphisms differentially influence the risk of gastric cancer in East Asians and Caucasians Cytokine 51 73–77 https://doi.org/10.1016/j.cyto.2010.03.007 PMID: 20363151
48. Kim J (2014) Ethnic differences in gastric cancer genetic susceptibility: allele flips of interleukin gene World J Gastroenterol 20 4558–4565 https://doi.org/10.3748/wjg.v20.i16.4558 PMID: 24782608 PMCID: 4000492
49. Garza E, Bosques F, and El-Omar E, et al (2005) Role of the polymorphic IL-1β, IL-1RN and TNF-α genes in distal gastric cancer in Mexico Int J Cancer 114 237–241 https://doi.org/10.1002/ijc.20718
50. Zhou Y, Li N, and Zhuang W, et al (2008) Interleukin-10-1082 promoter polymorphism associated with gastric cancer among Asians Eur J Cancer 44 2648–2654 https://doi.org/10.1016/j.ejca.2008.07.017 PMID: 18707865




