Brachytherapy reirradiation for local recurrences of gynaecological cancers: two clinical cases, literature review and OAR constraints
Javier Faundez1, Carlos Schananier2, Cynthia Villegas1 and Javier Retamales3
1Instituto Nacional del Cancer, Santiago 8380455, Chile
2Facultad de Medicina, Universidad de Chile, Santiago 8380453, Chile
3Hospital Sotero del Rio, Santiago 8207257, Chile
Abstract
Background: Local recurrence of gynaecological malignancies after prior pelvic radiotherapy presents a complex therapeutic dilemma. Pelvic exenteration is curative in select cases but carries high morbidity and profound quality-of-life impact. Salvage reirradiation with image-guided brachytherapy (BT) offers a highly conformal alternative; however, data remain fragmented and practical organ-at-risk (OAR) constraints are not standardised.
Objectives: 1) To describe in detail two contemporary cases of high-dose-rate (HDR) BT reirradiation – one vulvar and one vaginal vault recurrence – highlighting planning strategy, catheter implantation, dosimetry and clinical outcome; 2) to synthesise current evidence on efficacy, toxicity, dose–fractionation and technique of BT reirradiation in cervical, endometrial and vulvar cancers; and 3) to propose pragmatic cumulative EQD2 OAR limits for rectum, bladder, small bowel and urethra based on published series.
Methods: Both patients underwent computed tomography/magnetic resonance imaging-based contouring with hybrid interstitial or surface mould applicators. Total doses were 45.6 Gy in 8 fractions (EQD210 ≈ 60 Gy) for the vulvar recurrence and 24.6 Gy in 3 fractions (EQD24.5 ≈ 48 Gy) for the vaginal vault recurrence, with cumulative OAR doses referenced against literature-derived thresholds. A narrative review (1993–2024) identified 35 key studies involving >600 reirradiated patients.
Results: The vulvar case achieved complete response at 6 months without Grade ≥3 toxicity; the vaginal vault case showed initial partial response, remained ECOG 0 and required systemic therapy for later progression. Across the literature, 1- to 3-year local control ranged 40%–80% and overall survival 40%–60%, with severe late toxicity generally ≤30% when cumulative doses respected D2cc ≈ 70–100 Gy3 (rectum), 90–120 Gy3 (bladder) and D0.5cc ≤ 110 Gy3(urethra). Fractionation of 4–6 Gy BID for 5–10 fractions (EQD210 ≈ 40–50 Gy) was most common.
Conclusion: HDR BT reirradiation can achieve meaningful, sometimes durable, local control in carefully selected gynaecologic-cancer recurrences while preserving pelvic organs. Adhering to proposed cumulative EQD2 OAR constraints and employing image-guided interstitial techniques are paramount to curtail severe complications. Multidisciplinary assessment and treatment at experienced centres are strongly recommended to optimise outcomes.
Keywords: brachytherapy, salvage treatment, gynaecological cancer, narrative review
Correspondence to: Javier Faundez
Email: javier.faundez@incancer.cl
Published: 12/03/2026
Received: 29/05/2025
Publication costs for this article were supported by ecancer (UK Charity number 1176307).
Copyright: © the authors; licensee ecancermedicalscience. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Local recurrences of gynaecological malignancies – particularly cervical, endometrial and vulvar cancers – pose significant therapeutic challenges. Patients frequently have undergone extensive treatment, including radical surgery, external beam radiotherapy (EBRT) and sometimes chemotherapy. For many, the recurrent disease is confined to the central pelvis or vaginal vault. The traditional salvage procedure for central pelvic recurrence has often been pelvic exenteration. While this surgery may achieve local control, it is associated with high rates of morbidity and a profound impact on quality of life [1–3].
An alternative salvage approach is reirradiation, potentially using brachytherapy (BT). BT can deliver high, localised doses to the recurrent lesion while limiting radiation exposure to surrounding healthy structures. This targeted dose distribution is especially valuable in previously irradiated patients, for whom tolerance of normal tissues is often the limiting factor.
Although reirradiation with BT is not novel – historical case series date back decades [4] – its routine incorporation into practice has been slower, in part due to limited awareness, lack of training in advanced implant techniques and concerns about toxicity. Nonetheless, increasing availability of modern imaging computed tomography (CT) and magnetic resonance imaging (MRI) for treatment planning and advanced applicator systems has refined BT’s precision, making salvage reirradiation a viable option for many patients who may not tolerate or be candidates for surgery.
This article reports two clinical cases of salvage reirradiation in recurrent gynaecological cancers. It also summarises relevant literature that examines outcomes, toxicity*, dose considerations and technical aspects of image-guided brachytherapy for reirradiation. While the data are largely drawn from retrospective series, results suggest that reirradiation with BT can lead to substantial rates of local control – sometimes exceeding 50% – with acceptable late toxicity (mostly ≤ Grade 2), especially when delivered at experienced centers.
*All toxicities reported in this article are graded according to CTCAE v6.0
Case 1
History and presentation
An 86-year-old woman with a history of atrial fibrillation on direct oral anticoagulation presented initially with a vulvar tumour that extended across the left labium majus, the upper third of the right labium majus and the clitoris. Physical exam revealed a 3 cm fixed left inguinal node. A staging CT scan demonstrated diffuse thickening of both labia majora and a 3.4 cm left inguinal lymph node. Biopsy confirmed a moderately differentiated infiltrating squamous cell carcinoma, leading to a diagnosis of Stage IIIB vulvar cancer.
She received neoadjuvant EBRT (total of 47 Gy to the vulva and regional nodes and 58.8 Gy as a simultaneous integrated boost to the inguinal node) plus weekly cisplatin. Three months after completion, she underwent a radical vulvectomy and bilateral inguinal lymphadenectomy, with pathology showing no residual disease (pathologic complete response).
After more than 2 years of follow-up, a recurrent 4 × 2 cm vulvar tumour appeared at the labia majora and clitoris, sparing the urethra, anus and perineum. MRI suggested tumour abutting the distal urethra. A restaging CT showed no metastatic disease. As salvage surgery was deemed infeasible by the gynaecologic oncologist, the patient was referred for reirradiation with interstitial BT.
Treatment planning and procedure
A key step was a detailed clinical exam to determine the extent of disease and the feasibility of placing interstitial catheters at a safe distance from the urethra. A planning CT (2.5 mm slices) was fused with a diagnostic MRI to precisely delineate the gross tumour volume (GTV) (Figure 1).
Because the patient had previously received 47 Gy to the vulva (plus a higher boost to the lymph node area, which was anatomically separate), the oncologist and dosimetrist considered the dose already delivered to local organs at risk (OAR). The urethra, in particular, required careful attention. A common threshold is to maintain a cumulative dose of roughly 70–120 Gy EQD2_3 for the urethra, depending on the guidelines used [5].
In the pre-planning phase, an 8-fraction schedule (5.7 Gy/fraction) was proposed, for a total biologically effective dose of about 59.7 Gy EQD2_10. A cylinder placed in the vaginal canal was initially considered, but imaging showed that the recurrent vulvar lesion lay at a distance such that the cylinder would not effectively contribute dose. Instead, the plan required six to seven interstitial catheters.
Under spinal anesthesia, the implant procedure involved placing three flexible catheters on each side of the urethra at different depths (roughly 1 cm between each), plus an additional superficial catheter in the central region of the tumour. The catheters were secured with small radiopaque fixation buttons (Figure 2). A post-implant CT scan was performed to confirm the positioning of the catheters relative to the GTV and OAR.
The initial plan suggested possible undercoverage in superficial regions of the tumour (Figure 3). Therefore, a custom silicone bolus containing four additional catheters was placed on the vulvar surface to enhance coverage of peripheral edges. A repeat CT confirmed improved D100 and D90 of the GTV while maintaining urethral dose constraints (Table 1, Figure 4).
Outcomes and follow-up
Treatment was delivered over 1 week, with the patient hospitalised. She received eight fractions of high-dose-rate (HDR) BT (using an Ir-192 source). The urethra’s partial volumes stayed well below the established dose limit. Mild vulvar pain (grade 1) and grade 1 radiodermatitis were the main acute toxicities; both resolved within weeks. No acute or late toxicity was observed involving the rectum, bladder, small bowel or urethra. Toxicities were graded according to CTCAE v5.0 [30].
At 3 months, MRI and physical exam showed no evidence of residual disease. Six months post-treatment, she remained asymptomatic (ECOG 0), with no sign of local recurrence (Figure 5).
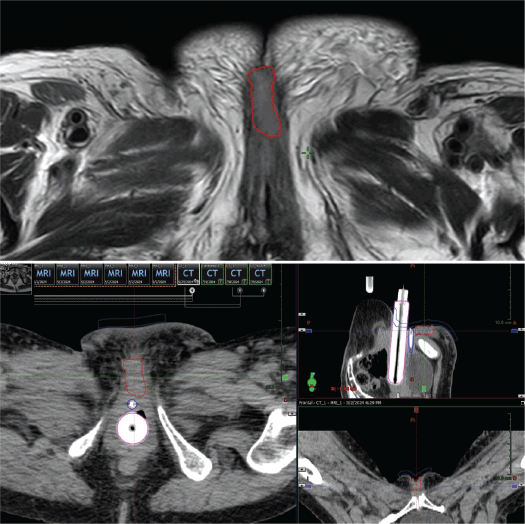
Figure 1. Case 1. Above, MRI of the pelvis, T2 sequence. The red line shows the GTV. Below, CT simulation for pre-plan design, on the axial, coronal and sagittal axis. The GTV is shown in red, the urethra in blue and the vaginal cylinder in pink.
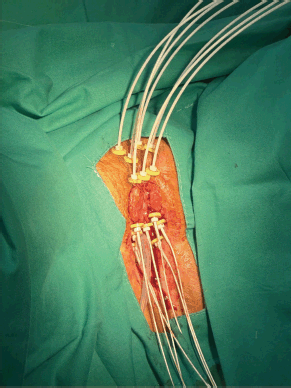
Figure 2. Case 1. The vulvar tumour and the implantation of seven interstitial catheters are observed.
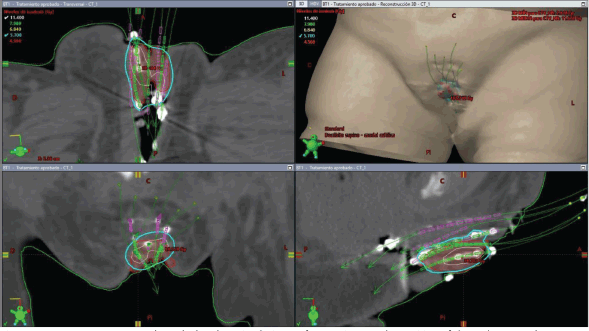
Figure 3. Case 1. 3D treatment plan calculated on simulation CT for fraction #1. The position of the catheters and the dosimetric distribution in the axial, coronal and sagittal planes are observed, in addition to a 3D reconstruction. In light blue, the isodose of 5.7 Gy; in white, the isodose of 11.4 Gy.
Table 1. Proposed OAR dose constraints in pelvic reirradiation. Dosimetric data from both clinical cases are shown. All doses are shown in EQD2, α/β = 3.
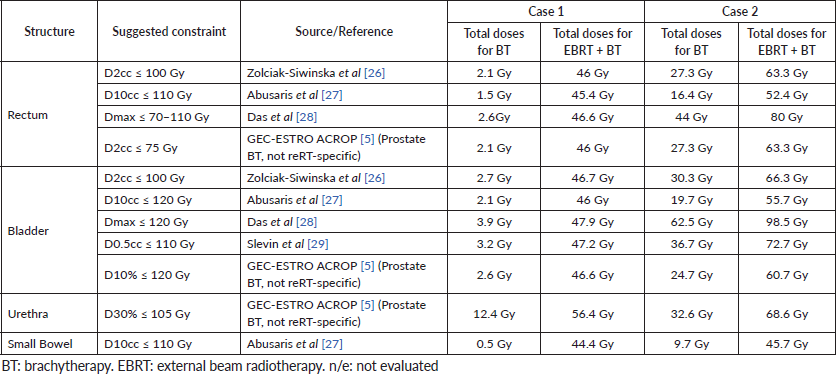
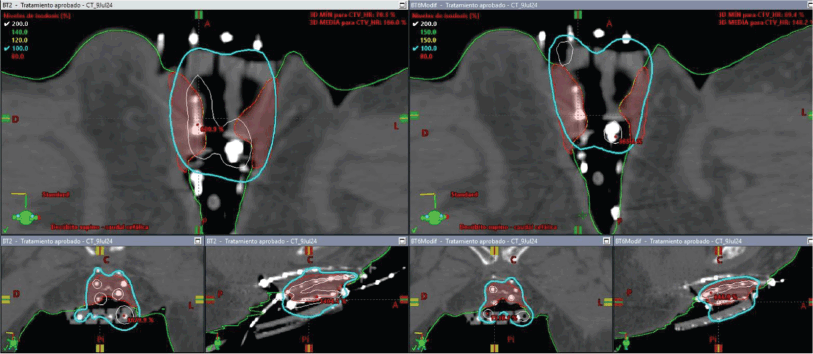
Figure 4. Case 1. 3D treatment plan with bolus. The position of the catheters and the dosimetric distribution in the axial, coronal and sagittal planes are observed. On the left, fraction #2 to #5. On the right, fraction #6 to #8. In light blue, the isodose of 5.7 Gy; in white, the isodose of 11.4 Gy.

Figure 5. Case 1. (a): Vulvar tumour prior to treatment. (b): 6 weeks later. (c): 3 months later. (d): 6 months later.
Case 2
History and presentation
A 75-year-old with a history of hypertension and dilated cardiomyopathy was diagnosed with Stage IIIC2 endometrial adenocarcinoma after hysterectomy, bilateral salpingo-oophorectomy and lymph node dissection revealed one positive para-aortic node. She declined or missed timely adjuvant radiotherapy. Seven months after surgery, the patient presented with a symptomatic recurrence in the vaginal vault and a positron emission tomography - computed tomography (PET-CT) also revealed latero-aortic hypermetabolic adenopathy. She received letrozole for hormone receptor–positive disease and six cycles of palliative chemotherapy with Carboplatin – Paclitaxel, after which she had complete regression of the adenopathy and stability of the disease in the vaginal vault. Then she was treated with palliative EBRT (30 Gy in 10 fractions) to the pelvis and vaginal vault. One year after this treatment, she developed progression of local disease involving the distal vagina and possibly the urethra (Figure 6). The case was discussed in tumour board, and pelvic exenteration was considered but deemed too morbid. She was referred for HDR interstitial BT reirradiation.
Planning and implant
Because her tumour extended within the entire vaginal canal, with possible urethral involvement, a hybrid endocavitary–interstitial approach was chosen. A custom 3D-printed template was designed with multiple channels to guide needle insertion transperineally, along with a central opening for a 2 cm cervical applicator.
On the day of implant, the patient underwent spinal anesthesia, and a Foley catheter was placed. The custom template was positioned against the perineum, and 18 interstitial needles were inserted according to the pre-plan. An initial fraction was delivered at 8.2 Gy to the 90% isodose of the GTV (Figure 7). For subsequent fractions, the central channel was exchanged for a deeper needle to cover more cranial disease.
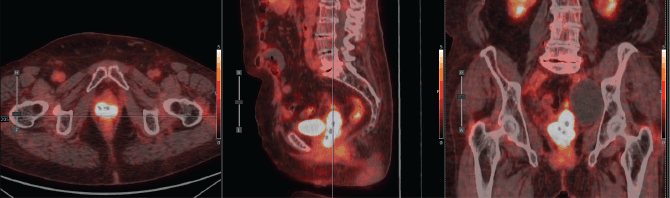
Figure 6. Case 2. PET-CT scan prior to BT. Axial, sagittal and coronal section. A hypermetabolic tumour mass in the vaginal vault and a left pelvic lymphocele were observed.
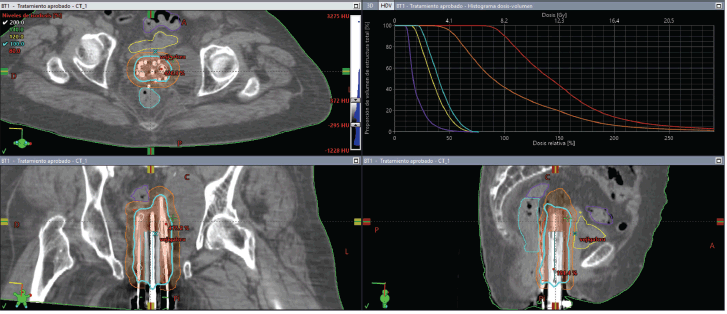
Figure 7. Case 2. Dose-volume histogram and dosimetric distribution of fraction #1 in axial, coronal and sagittal planes. It is shown in red CTV-HR, in orange CTV-IR, in light blue the rectum, in yellow the bladder and in purple the sigmoid colon.
Total dose was 8.2 Gy × 3 fractions, equating to roughly 48.1 Gy EQD24.5. The plan accounted for prior EBRT (30 Gy in 10 fractions). Although the bladder, rectum and sigmoid colon had some overlap with the new target volume, no dose-limiting constraints were exceeded (Table 1).
Outcomes and follow-up
The patient developed transient Grade 2 urinary irritative symptoms (frequency and dysuria) that resolved within 4 weeks post-treatment. No rectal bleeding, bowel changes or urethral discomfort were reported. No late Grade ≥2 toxicities involving the rectum, bladder, small bowel or urethra were documented. All toxicities were assessed and graded per CTCAE v5.0. Four months later, a PET-CT showed a slight decrease in tumour size and metabolism in the vault and proximal vaginal canal, measuring 40 mm (versus 48 mm on the pre-treatment PET-CT) and an SUVmax of 11 (versus 18). Because of this residual tumour activity, the patient received a second course of palliative chemotherapy with Carboplatin – Paclitaxel for six cycles. New images showed local and systemic progression. Doxorubicin was started then. Nonetheless, from a performance standpoint, she remained ECOG 0 after BT, capable of normal daily activities. Twenty-one months post-BT, she continued palliative treatment with no major local complications attributed to reirradiation.
Literature review
BT allows very focal dose escalation in sites where EBRT alone might expose the bowel, bladder or other pelvic structures to unsafe levels [4, 6]. A recurrent lesion often lies in the central pelvis or vaginal vault. If surgery is not feasible, reirradiation is one of the few remaining curative-intent options.
While randomised trials on reirradiation in gynaecological cancer are extremely scarce, numerous retrospective series underscore the feasibility and effectiveness of this approach in carefully selected patients. Overall local control rates vary between 40% and 80% depending on tumour size, prior dose, site of recurrence and technique [7–9].
In a prospective phase II trial, Martínez-Monge et al [7] analysed 50 patients with locally advanced and recurrent gynaecological cancer – 25 of whom had received prior irradiation. In patients without previous radiation, the treatment approach consisted of neoadjuvant chemoradiotherapy followed by surgery and HDR BT, whereas in those who had already been irradiated, surgery was combined with HDR BT. Long-term follow-up (median 11.5 years) demonstrated a local control rate of 59.6% at 14 years among the reirradiated cohort, although dose reductions became necessary to reduce Grade ≥3 toxicities. Another study by Amsbaugh et al [8], retrospectively evaluating 21 patients with recurrent gynaecologic cancers who underwent interstitial BT (median total dose of 22.5 Gy in HDR or approximately 41.5 Gy in low-dose-rate (LDR)), noted a 1-year locoregional failure-free rate of roughly 71.5% and an 82.2% 1-year overall survival (OS), with Grade 3 or higher vaginal, urinary or rectal toxicities staying below 30%. Similarly, Kellas-Ślęczka et al [9] assessed interstitial HDR BT for 14 patients with locally advanced or recurrent vulvar cancer and observed favourable local control rates alongside minimal Grade 3–4 toxicities. A broad review by Sturdza et al [10] (published under the auspices of the American BT Society working group), examining reirradiation for cervical and endometrial cancers, underscored that BT alone or in conjunction with a more limited EBRT field can offer curative reirradiation for small recurrences, provided normal tissue constraints are respected.
Focusing on recurrent endometrial cancer, Ling et al [11] documented a 66% 3-year local control rate in 22 patients reirradiated with three-dimensional BT, with or without EBRT; many of these individuals had previously received either EBRT or vaginal BT. Huang et al [12], investigating a cohort of 16 patients with recurrent endometrial cancer, reported that individualised reirradiation doses based on risk categories yielded a 53% local control rate at 2 years, although four patients experienced some degree of toxicity. In the context of recurrent cervical cancer, Mahantshetty et al [13] evaluated 30 patients who were reirradiated using interstitial or intracavitary HDR BT, finding a 2-year local control rate of 44% overall but 52% in those receiving higher doses (exceeding 40 Gy EQD2). Likewise, Mabuchi et al [15] examined a group of 52 patients with central recurrences who were administered 6 Gy × 7 fractions, noting a 77% clinical response at 2 months, though around 25% of patients experienced severe toxicity. LDR or permanent implants, such as 198Au or 131Cs, have also shown promise, with local control rates of about 70%–85% for small or superficially located recurrences [16–19].
Regarding dose and fractionation regimens, HDR BT frequently employs 4–6 Gy per fraction administered twice daily, targeting an overall EQD2_10 of 40–50 Gy or more, though once-weekly high-dose treatments have also been described. The decision on fractionation schedule typically hinges on tumour size, OAR proximity, previous radiation dose and the time elapsed since earlier radiotherapy. Some centers opt for more conservative total doses – around 24–32 Gy – to curtail severe toxicity, but there is a potential reduction in local control if the total dose is insufficient [7, 10]. In certain cases, stereotactic body radiotherapy (SBRT) is used for pelvic reirradiation, but substantial toxicity, such as bowel injury or fistula formation, remains a concern in extensively pretreated fields [20, 21]. If prior irradiation was restricted to the vaginal vault, external beam reirradiation can sometimes be extended to incorporate the pelvis, sparing previously unirradiated areas, though full-dose salvage across large volumes may be challenging [24].
Discussion
Patient selection and prescription dose proposal
Candidates for salvage reirradiation must have a good performance status (ECOG 0–2) and no evidence of extensive metastatic disease. They should ideally have a tumour size amenable to BT coverage – often up to ~4–5 cm in diameter – though some larger lesions have been tackled with combined interstitial and intracavitary techniques. Adequate healing capacity and acceptance of possible complications are also critical.
All patients should be evaluated in a multidisciplinary tumour board to ensure optimal treatment selection. BT reirradiation should be considered in patients for whom surgery is contraindicated due to significant comorbidities or in those who explicitly decline surgical management. It is particularly valuable in clinical scenarios where pelvic exenteration is deemed excessively morbid, allowing for a potentially equivalent rate of local control with substantially lower morbidity.
From the perspective of the radiation oncologist, once the above criteria are met, the decision to proceed with reirradiation should follow a detailed pre-planning process. This includes summing the dose contributions from the prior course and estimating the cumulative dose to OARs based on the expected distribution of the new treatment. We recommend considering a prescription dose in the range of 40–47.5 Gy EQD2₁₀ as the minimum necessary to achieve effective tumour control, as supported by the evidence summarised in Table 2.
An interval of at least 12 months between previous irradiation and the new course is often cited as a factor for lower toxicity [25]. Some authors note that patients with intervals under 1 year can still be treated, but with an elevated risk of complications.
Technique considerations
Modern BT relies heavily on imaging guidance (CT, MRI) to place catheters or needles precisely. Interstitial HDR BT is particularly beneficial for irregular, bulky or lateral recurrences, as the catheters can be placed directly in or around the tumour. Hybrid techniques combine intracavitary (e.g., tandem, vaginal cylinders) with needle implants for better coverage.
Dosimetry commonly focuses on ensuring the prescription dose (e.g., 90% isodose coverage of the CTV) while keeping OAR volumes below defined thresholds. The dose-limiting structures often include the urethra (for vulvar or distal vaginal recurrences), bladder and rectum.
Outcomes and prognostic factors
In broad retrospective data, salvage BT reirradiation can yield local control rates from about 40% to over 80%, depending on tumour size, total dose and the interval since previous radiation [7–10]. Smaller tumours, higher prescription doses and longer radiation-free intervals tend to correlate with improved outcomes.
Table 2. Summary of studies on BT-based reirradiation in gynaecologic cancer.
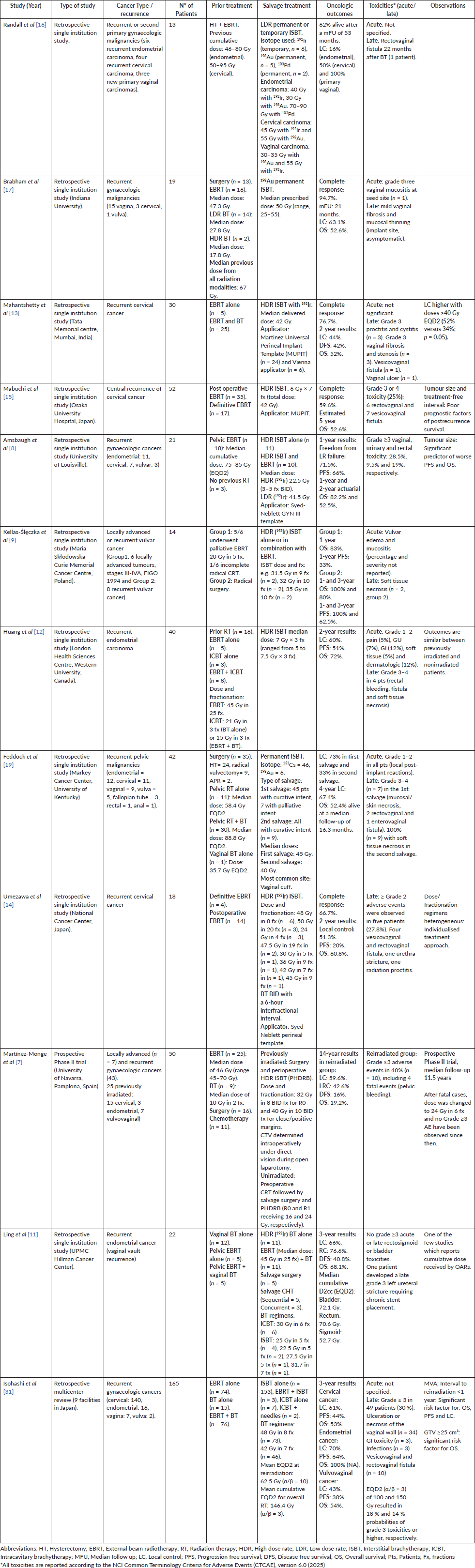
Severe (grade 3–4) late toxicities, such as fistulas (rectovaginal, vesicovaginal), necrosis or severe bleeding, remain a risk but can be mitigated by meticulous planning and careful fractionation. Some reports suggest higher toxicities when total doses exceed 40–50 Gy EQD2_10, particularly in previously irradiated patients [7, 15]. However, it is a balance: too low a dose may fail to control the recurrence.
Comparison with surgery
For central pelvic recurrences, exenteration can be curative. However, exenteration entails extensive morbidity, reduced quality of life and a prolonged recovery. The 5-year OS can range from 10% to 56%, with perioperative morbidity rates of 22%–66% [10, 23]. By contrast, salvage BT can preserve organ function and potentially avoid major surgery, though it must be carefully weighed against the risk of complications in tissues already near tolerance.
Role of systemic therapy
Concurrent chemotherapy is sometimes added to reirradiation protocols, though there is no universal consensus. Some authors prefer sequential or neoadjuvant chemotherapy [8, 22], aiming to shrink the tumour before BT. Others worry about increased risks of fistula formation, especially when combining radiosensitising chemotherapy with a high-dose local approach. Decisions tend to be individualised.
Proposed OAR constraints for pelvic reirradiation
In reirradiation scenarios, tolerance of OAR is the primary dose-limiting factor. Because the pelvis has often received substantial prior exposure, cumulative doses can rapidly approach or exceed tissue tolerance. Several authors have proposed guidelines for rectum, bladder, small bowel and urethra based on retrospective analyses of reirradiation in various settings (e.g., BT, SBRT). While there is no universal consensus, the values presented below in Table 1 serve as a useful reference. In practice, clinicians should adapt these constraints to each patient’s prior dose distribution, the time elapsed since previous irradiation and specific technique (e.g., HDR BT versus external beam). In some cases, guidelines originally developed for prostate BT (e.g., GEC-ESTRO ACROP) are adapted cautiously for gynaecological reirradiation, with the understanding that the clinical context differs.
Practical considerations and future directions
In many institutions, the number of annual reirradiation cases is relatively small [10]. This underscores the importance of centralised care or referral to centers with significant expertise in BT for recurrent disease. Standardising OAR constraints, prescribing guidelines and best practices for needle implant techniques could improve outcomes further.
Ongoing developments in 3D printing have fostered patient-specific applicators, as illustrated in Case 2. Such approaches permit more personalised implantation and coverage of anatomically challenging regions. The routine use of MRI-based planning also refines delineation of the target and healthy structures, potentially reducing margins while ensuring robust coverage.
Prospective trials in this area remain difficult, given the heterogeneity of recurrent disease, prior therapy and patient comorbidities. Nonetheless, collaborative research and registries can help refine dose constraints, define fractionation schedules and identify prognostic factors that predict for cure versus high toxicity.
Conclusion
Reirradiation with BT is an important salvage approach for local recurrences of gynaecological malignancies in patients who cannot undergo or decline radical surgery. Modern imaging and implant techniques have helped reduce toxicity while improving local control rates, which, in well-selected patients, can be comparable to or better than surgical outcomes.
The two clinical cases presented illustrate the process of careful patient selection, detailed pre-planning to ensure feasibility, implant procedures tailored to each anatomical site and attention to cumulative OAR doses. Despite differences in disease site (vulva versus vaginal vault), both patients tolerated the salvage procedure without severe toxicity. One achieved a complete response at 6 months, while the other had an initial partial response, later requiring additional systemic therapy for progressive disease.
A robust body of retrospective evidence supports the potential curative role of BT-based reirradiation, especially for relatively small, central or paracentral recurrences. Adequate performance status, acceptable prior dose levels and at least 1 year since previous radiation are important selection factors. Adherence to cumulative dose constraints – particularly to the rectum, bladder and urethra – can reduce the risk of life-altering complications like fistulas.
While many questions remain regarding optimal fractionation, total dose and the role of concurrent chemotherapy, BT stands as a vital tool in the armamentarium against recurrent gynaecological cancers. As imaging and treatment planning technology continue to evolve, it is likely that reirradiation with BT will become increasingly safe, precise and effective for the challenging scenario of local relapse in already-irradiated tissues.
Conflicts of interest
There are no conflicts of interest for any of the authors.
Funding
No funding was provided for this manuscript.
References
1. Maggioni A, Roviglione G, and Landoni F, et al (2009) Pelvic exenteration: ten-year experience at the European Institute of Oncology in Milan Gynecol Oncol 114 64–68 https://doi.org/10.1016/j.ygyno.2009.03.029 PMID: 19411097
2. Berek JS, Howe C, and Lagasse LD, et al (2005) Pelvic exenteration for recurrent gynecologic malignancy: survival and morbidity analysis of the 45-year experience at UCLA Gynecol Oncol 99 153–159 https://doi.org/10.1016/j.ygyno.2005.05.034 PMID: 16054678
3. Miller B, Morris M, and Rutledge F, et al (1993) Aborted exenterative procedures in recurrent cervical cancer Gynecol Oncol 50 94–99 https://doi.org/10.1006/gyno.1993.1170 PMID: 8349169
4. Nag S, Selman AE, and Martinez-Monge R (1998) Interstitial reirradiation as salvage treatment for previously irradiated patients with recurrent gynecologic malignancies J Brachytherapy Int 14 49–57
5. Henry A, Pieters BR, and André Siebert F, et al (2022) GEC-ESTRO ACROP prostate brachytherapy guidelines Radiother Oncol 167 244–251 https://doi.org/10.1016/j.radonc.2021.12.047 PMID: 34999134
6. De Boer SM, Powell ME, and Mileshkin L, et al (2018) Adjuvant chemoradiotherapy versus radiotherapy alone for high-risk endometrial cancer (PORTEC-3) Lancet Oncol 19(3) 295–309 https://doi.org/10.1016/S1470-2045(18)30079-2 PMID: 29449189 PMCID: 5840256
7. Martínez-Monge R, Valtueña Peydró G, and Cambeiro M, et al (2018) Perioperative high-dose-rate brachytherapy in locally advanced and recurrent gynecological cancer: final results of a Phase II trial Brachytherapy 17(5) 734–741 https://doi.org/10.1016/j.brachy.2018.04.007 PMID: 29803537
8. Amsbaugh MJ, Bhatt N, and Hunter T, et al (2015) Computed tomography planned interstitial brachytherapy for recurrent gynecologic cancer Brachytherapy 14(5) 600–605 https://doi.org/10.1016/j.brachy.2015.05.007 PMID: 26087868
9. Kellas-Ślęczka S, Białas B, and Fijałkowski M, et al (2016) Interstitial high-dose-rate brachytherapy in locally advanced and recurrent vulvar cancer J Contemp Brachytherapy 1 32–40 https://doi.org/10.5114/jcb.2016.58081
10. Sturdza A, Viswanathan AN, and Erickson B, et al (2020) American Brachytherapy Society working group report on the patterns of care and a literature review of reirradiation for gynecologic cancers Brachytherapy 19(2) 127–138 https://doi.org/10.1016/j.brachy.2019.11.008 PMID: 31917178
11. Ling DC, Vargo JA, and Glaser SM, et al (2019) Outcomes after definitive re-irradiation with 3D brachytherapy with or without external beam radiation therapy for vaginal recurrence of endometrial cancer Gynecol Oncol 152 581–586 https://doi.org/10.1016/j.ygyno.2018.12.022 PMID: 30600093 PMCID: 7422921
12. Huang K, D'Souza D, and Patil N, et al (2016) High-dose-rate interstitial brachytherapy for the treatment of high-volume locally recurrent endometrial carcinoma Brachytherapy 15 543–548 https://doi.org/10.1016/j.brachy.2016.06.010 PMID: 27475479
13. Mahantshetty U, Kalyani N, and Engineer R, et al (2014) Reirradiation using high-dose-rate brachytherapy in recurrent carcinoma of uterine cervix Brachytherapy 13 548–553 https://doi.org/10.1016/j.brachy.2014.06.005 PMID: 25085457
14. Umezawa R, Murakami N, and Nakamura S, et al (2018) Image-guided interstitial high-dose-rate brachytherapy for locally recurrent uterine cervical cancer Brachytherapy 17 368–376 https://doi.org/10.1016/j.brachy.2017.11.011
15. Mabuchi S, Takahashi R, and Isohashi F, et al (2014) Reirradiation using high-dose-rate interstitial brachytherapy for locally recurrent cervical cancer Int J Gynecol Cancer 24 141–148 https://doi.org/10.1097/IGC.0000000000000028
16. Randall ME, Evans L, and Greven KM, et al (1993) Interstitial reirradiation for recurrent gynecologic malignancies: results and analysis of prognostic factors Gynecol Oncol 48 23–31 https://doi.org/10.1006/gyno.1993.1005 PMID: 8423018
17. Brabham JG and Cardenes HR (2009) Permanent interstitial reirradiation with 198Au as salvage therapy for low volume recurrent gynecologic malignancies: a single institution experience Am J Clin Oncol 32 417–422 https://doi.org/10.1097/COC.0b013e318191bfc7 PMID: 19451799
18. Wooten CE, Randall M, and Edwards J, et al (2014) Implementation and early clinical results utilizing Cs-131 permanent interstitial implants for gynecologic malignancies Gynecol Oncol 133 268–273 https://doi.org/10.1016/j.ygyno.2014.02.015 PMID: 24556059
19. Feddock J, Cheek D, and Steber C, et al (2017) Reirradiation using permanent interstitial brachytherapy: a potentially durable technique for salvaging recurrent pelvic malignancies Int J Radiat Oncol Biol Phys 99 1225–1233 https://doi.org/10.1016/j.ijrobp.2017.08.027 PMID: 29029888
20. Yazici G, Sari SY, and Yedekci FY, et al (2016) The dosimetric impact of implants on the spinal cord dose during stereotactic body radiotherapy Radiat Oncol 11 71 https://doi.org/10.1186/s13014-016-0649-z PMID: 27225270 PMCID: 4880816
21. Pontoriero A, Iati G, and Aiello D, et al (2016) Stereotactic radiotherapy in the retreatment of recurrent cervical cancers, assessment of toxicity, and treatment response: initial results and literature review Technol Cancer Res Treat 15 759–765 https://doi.org/10.1177/1533034615608740
22. Moschiano EJ, Barbuto DA, and Walsh C, et al (2014) Risk factors for recurrence and prognosis of low-grade endometrial adenocarcinoma; vaginal versus other sites Int J Gynecol Pathol 33 268–273 https://doi.org/10.1097/PGP.0b013e31829c6757 PMID: 24681738
23. Bockel S, Espenel S, and Sun R, et al (2021) Image-Guided Brachytherapy for Salvage Reirradiation: a Systematic Review Cancers 13(6) 1226 https://doi.org/10.3390/cancers13061226 PMID: 33799617 PMCID: 7999189
24. Lancellotta V, Macchia G, and Garganese G, et al (2021) The role of brachytherapy (interventional radiotherapy) for primary and/or recurrent vulvar cancer: a Gemelli Vul.Can multidisciplinary team systematic review Clin Transl Oncol 23(8) 1611–1619 https://doi.org/10.1007/s12094-021-02557-1 PMID: 33650029
25. Llewelyn M and Taylor A (2017) Re-irradiation of cervical and endometrial cancer Curr Opin Oncol 29(5) 343–350 https://doi.org/10.1097/CCO.0000000000000392 PMID: 28697002
26. Zolciak-Siwinska A, Bijok M, and Jonska-Gmyrek J, et al (2014) HDR brachytherapy for the reirradiation of cervical and vaginal cancer: analysis of efficacy and dosage delivered to organs at risk Gynecol Oncol 132 93–97 https://doi.org/10.1016/j.ygyno.2013.10.018
27. Abusaris H, Hoogeman M, and Nuyttens JJ (2012) Re-irradiation: outcome, cumulative dose and toxicity in patients retreated with stereotactic radiotherapy in the abdominal or pelvic region Technol Cancer Res Treat 11(6) 591–597 https://doi.org/10.7785/tcrt.2012.500261 PMID: 22568625 PMCID: 4527454
28. Mukherji A, Das S, and Patro K (2018) Recovery and tolerance of the organs at risk during reirradiation J Curr Oncol 1(1) 23 https://doi.org/10.4103/jco.jco_2_17
29. Slevin F, Aitken K, and Alongi F, et al (2021) An international Delphi consensus for pelvic stereotactic ablative radiotherapy re-irradiation Radiother Oncol 164 104–114 https://doi.org/10.1016/j.radonc.2021.09.010 PMID: 34560186
30. National Cancer Institute (2025) Common Terminology Criteria for Adverse Events (CTCAE) version 6.0 Bethesda (MD): U.S. Department of Health and Human Services [https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events]
31. Isohashi F, Yoshida K, and Murakami N, et al (2024) Reirradiation for recurrent gynecologic cancer using high-dose-rate brachytherapy in Japan: a multicenter survey on practice patterns and outcomes Radiother Oncol 195 110269 https://doi.org/10.1016/j.radonc.2024.110269 [https://www.sciencedirect.com/science/article/pii/S0167814024001919]





