Purpose: This phase 2 study evaluated the safety of adjuvant chemoradiation (CTRT) for breast cancer.
Methods: From April 2019 to 2020, 60 patients with stage II–III invasive breast cancer planned for adjuvant taxane-based chemotherapy and radiotherapy (RT) were accrued. Local ± regional (excluding the internal mammary nodal region) RT (40 Gy in 15 fractions ± boost) was started with the third cycle of an adjuvant taxane in a 3-weekly schedule or with the eighth cycle in a weekly schedule.
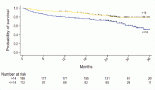
Results: Thirty-six patients received 3-weekly paclitaxel regimen and 24 received weekly paclitaxel regimen. The commonly used technique was three-dimensional conformal RT which was employed in 58% of patients. Regional RT, including the medial supraclavicular region, was done in 42 patients (70%). No dose-limiting (grade 3 or 4) toxicity was documented and all patients completed CTRT without any treatment interruption. The median ejection fraction pre and post CTRT 6 months was 60% (p = 0.177). The median value of cardiac enzyme (Troponin T ng/L) decreased from 37 to 20 (p = 0.009) post CTRT 6 months. Of the 54 patients who underwent the pulmonary function tests, there was no significant difference in various parameters like functional vital capacity (FVC) (2.29 versus 2.2 L, p = 0.375), forced expiratory volume at 1 second (FEV1) (1.86; 1.82; p = 0.365), FEV1/FVC (81.5; 81.43; p = 0.9) and diffusion lung capacity for carbon monoxide (88.3; 87.6; p = 0.62). At a median follow-up of 34 months, the 3-year actuarial rate of disease-free survival and overall survival was 75% and 98.3%, respectively. Quality of life scores (QOL) improved after treatment for most of the domains comparable to the pre-RT scores.
Conclusion: Taxane-based adjuvant CTRT is a safe option and results in minimal toxicity and excellent compliance. It has favourable impact on cardio-pulmonary profile and QOL scores.