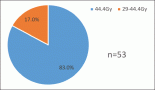
Azacitidine (AZA), as demonstrated in the phase III trial (AZA-001), is the first MDS treatment to significantly prolong overall survival (OS) in higher risk MDS pts ((2007) Blood 110 817). Approximately, one-third of the patients (pts) enrolled in AZA-001 were FAB RAEB-T (¡Ý20¨C30% blasts) and now meet the WHO criteria for acute myeloid leukaemia (AML) ((1999) Blood 17 3835). Considering the poor prognosis (median survival <1 year) and the poor response to chemotherapy in these pts, this sub-group analysis evaluated the effects of AZA versus conventional care regimens (CCR) on OS and on response rates in pts with WHO AML.